Now, we provide various types of extension sets, including iv extension set with injection port, iv extension set with luer lock, microbore extension set, small bore extension set, syringe extension set, y extension set, etc.
Introduction Of Extension Sets
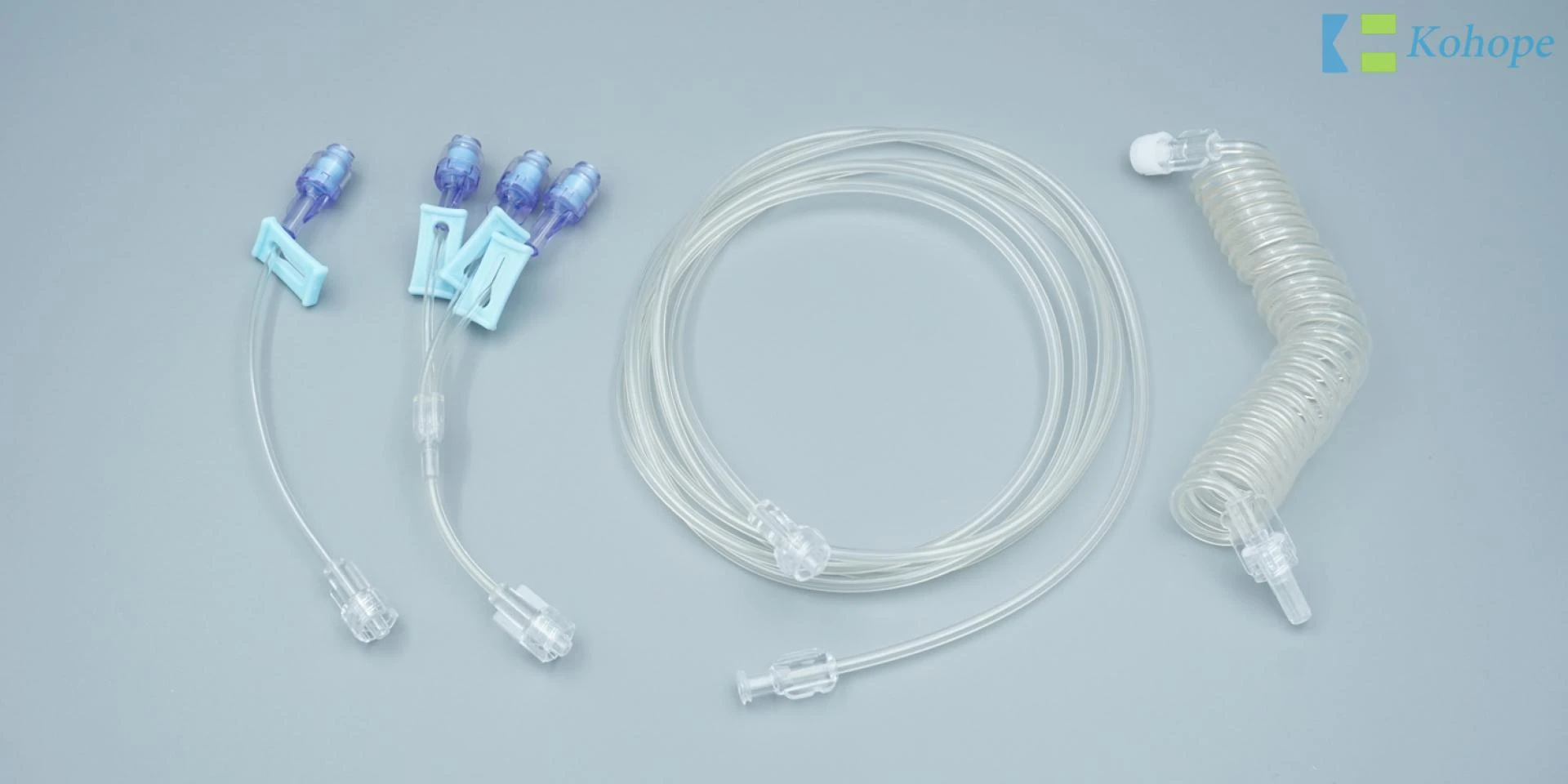
The extension sets are assembled by a protective cap, conical lock fitting. three-way connector, tubing and clip, and so on.
The catheter extension set is a kind of multifunction connection tube, can be widely used in infusion performance, for example, infusion container, infusion instrument, infusion sets. And the iv extension line also help to increase the medicine filter, flow speed adjustment, or adding medicine of increase set. Also the iv extension set can be used to add the length of the infusion tube.
Extension Sets One
Characteristic:
Male and female Luer adapter
Slide or lock clamp
Standard tubing
Y-injection site
0.2/1.2 micron filter
Flow controller
Sterile
Extension Sets Two
Characteristic:
Male and female Luer adapter
Slide or lock clamp
Standard tubing
Sterile
Connector
Characteristic:
Male and female Luer adapter
Slide or lock clamp
Standard tubing
Sterile
Y-injection Site
Characteristic:
Male and female Luer adapter
Slide or lock clamp
Standard tubing
Sterile
Mini Tubing
Characteristic:
Male and female Luer adapter
Slide or lock clamp
Mini tubing
Sterile
High-pressure Tubing
Characteristic:
Male and female Luer adapter
Slide or lock clamp
High-pressure tubing
Sterile
Details Of Extension Sets
Main material: PE, TPU, PVC, MBS
Characteristic:
Male and female Luer adapter
Slide or lock clamp
Standard tubing
Y-injection site
0.2/1.2 micron filter
Flow controller
Sterile
Other Notes Of Hypodermic Syringes.
Product Conformance:
Conform to ISO 8536-4, ISO8536-9
In compliance with European Medical Device Directive 93/42/EEC(CE Class: lla)
Quality Assurance:
The manufacturing process is in compliance with ISO 13485 and ISO 9001 Quality System.
How should we define a good disposable medical product? Is a good disposable medical product just refer to the good quality? Kohope Medical Devices Co., Ltd. would like to tell you that based on high quality, safety medical supplies also pursue a wonderful sense of use, convenient follow-up treatment, and environmental protection. Kohope Medical Devices Co., Ltd., as a hospital medical supplier, have found their balance point in the process of searching and finally brought you top-quality and functional medical disposable products.






Sign in to leave a comment.