In the fast-paced world of medical device development, integrating Agile methodologies with Change Control processes is crucial for ensuring product quality and regulatory compliance. This blog explores how Agile practices can enhance Change Control in Medical Devices, and provides a comprehensive guide on managing Change Management Processes effectively in this sector.
1. Understanding Change Control in Medical Devices
1.1 What is Change Control in Medical Devices?
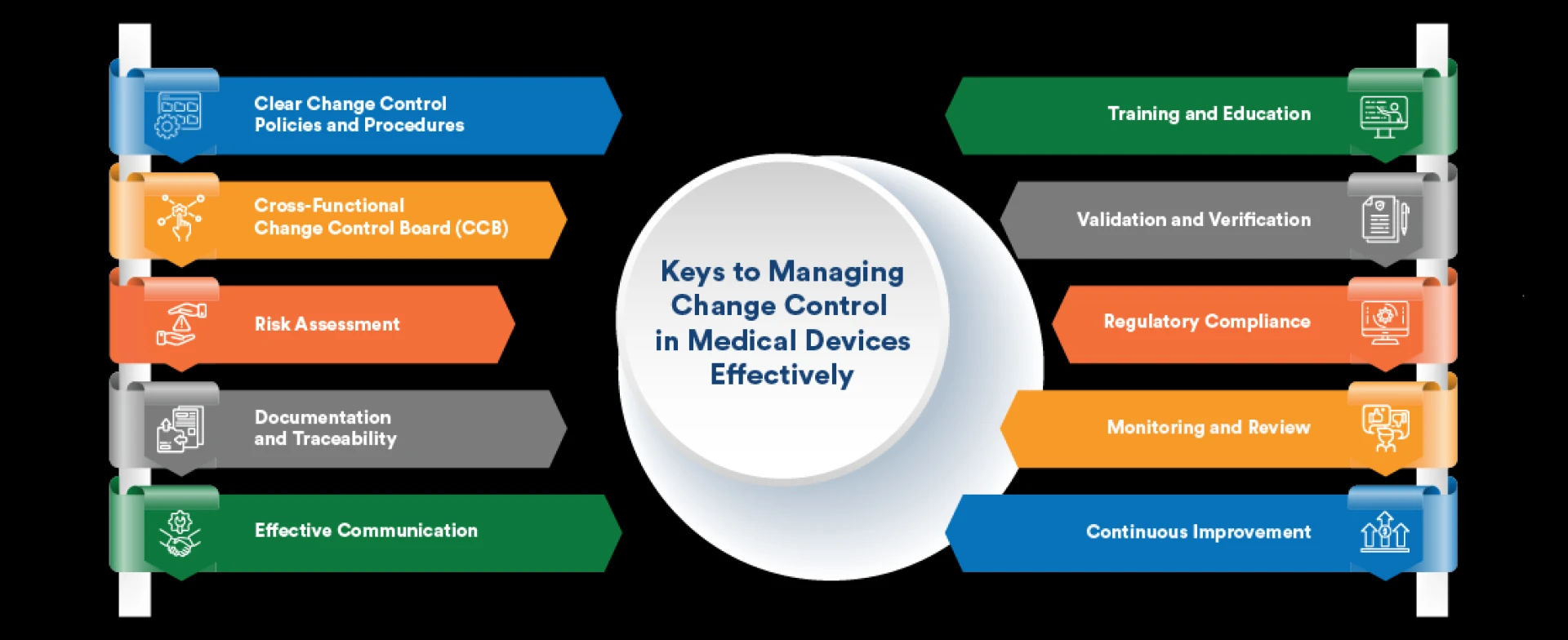
Change Control in Medical Devices involves managing and documenting changes to ensure that products meet quality and regulatory standards. This process is essential for maintaining compliance and ensuring safety and efficacy throughout the device lifecycle.
1.2 Importance of Effective Change Control
Effective Change Control helps prevent errors, ensures consistent product quality, and maintains regulatory compliance. It is critical for managing modifications in medical device projects, as any deviation can impact patient safety and device performance.
The Role of Agile Methodologies in Medical Device Projects
2.1 Overview of Agile Methodologies
Agile methodologies emphasize iterative development, collaboration, and flexibility. This approach allows teams to adapt quickly to changes, which is particularly beneficial in the dynamic field of medical device development.
2.2 Benefits of Agile in Medical Device Development
Implementing Agile in medical device projects offers several benefits, including improved responsiveness to market needs, increased collaboration, and faster time-to-market. Agile practices facilitate ongoing refinement and enhancement of products, which is essential for staying competitive.
3. Integrating Agile Practices with Change Control
3.1 How Agile Practices Enhance Change Control in Medical Devices
Integrating Agile practices with Change Control in Medical Devices allows for more adaptive and responsive change management. Agile practices support frequent updates and iterative improvements, aligning well with the needs of change control processes.
3.2 Agile Frameworks for Change Control
Different Agile frameworks, such as Scrum and Kanban, can be adapted to support Change Control processes. These frameworks provide structured approaches to managing changes while maintaining flexibility and responsiveness.
4. Implementing Agile Change Control in Medical Device Projects
4.1 Steps to Integrate Agile with Change Control
Assess Current Change Control Processes: Evaluate existing Change Control in Medical Devices to identify areas for improvement.Adopt Agile Frameworks: Choose an Agile framework that best fits your project needs.Train Teams: Ensure all team members are trained in Agile methodologies and their application to Change Control.Implement Agile Practices: Apply Agile practices to manage changes iteratively and collaboratively.4.2 Common Challenges and Solutions
Challenge: Resistance to ChangeSolution: Provide adequate training and support to facilitate smooth transitions.Challenge: Integration with Existing ProcessesSolution: Customize Agile practices to align with established Change Management Processes.
5. Case Studies: Successful Integration of Agile and Change Control
5.1 Case Study 1: Improving Efficiency in Medical Device Development
A leading medical device company integrated Agile methodologies with their Change Control processes, resulting in faster development cycles and improved product quality.
5.2 Case Study 2: Enhancing Regulatory Compliance
Another example highlights how Agile practices helped a company better manage compliance requirements and streamline their Change Management Process, ensuring adherence to regulations.
6. Best Practices for Agile Change Control in Medical Devices
6.1 Establish Clear Change Management Processes
Define and document Change Management Processes clearly to ensure consistency and compliance.
6.2 Foster Collaboration and Communication
Encourage open communication and collaboration among team members to facilitate effective change management.
6.3 Monitor and Adjust Agile Practices
Regularly review and adjust Agile practices to ensure they continue to meet the needs of Change Control in Medical Devices.
7. The Impact of CAPA on Change Control in Agile Projects
7.1 Understanding CAPA in Pharmaceutical Industry
CAPA in the Pharmaceutical Industry for addressing and preventing quality issues. Its principles are also applicable to Change Control in Medical Devices.
7.2 Applying CAPA Medical Device Principles
Incorporating CAPA Medical Device principles into Agile Change Control processes ensures that corrective actions are implemented effectively and preventive measures are in place.
8. Future Trends in Agile and Change Control Integration
8.1 Advancements in Agile Methodologies
Future advancements in Agile methodologies may further enhance their integration with Change Control processes, leading to more efficient and compliant medical device development.
8.2 Emerging Technologies and Their Impact
Emerging technologies, such as AI and machine learning, may offer new tools and techniques for managing changes in medical device projects.
Conclusion
Integrating Agile methodologies with Change Control in Medical Devices can significantly enhance project efficiency and compliance. To successfully manage this integration, utilizing advanced tools like ComplianceQuest Management Software is essential. ComplianceQuest provides a robust platform for managing Change Control, CAPA, and Change Management Processes, ensuring regulatory compliance and operational excellence. As the medical device industry continues to evolve, adopting such comprehensive solutions will be critical for staying competitive and meeting industry standards in 2024.







Sign in to leave a comment.