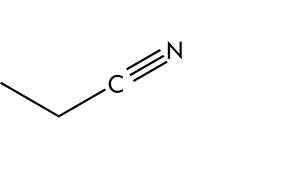
The chemical formula for propionitrile, sometimes referred to as ethyl cyanide, is CH3CH2CN. It is an odourless, colourless liquid that dissolves in water, ethanol, and ether. Propionitrile is largely employed as a solvent in the manufacture of agrochemicals, fragrances, and medicines.
The reactions of propionyl chloride and sodium cyanide, acrylonitrile and ethylene, and the dehydration of propanamide are only a few of the processes that may be used to make propionitrile. Additionally, acetonitrile can be catalytically hydrogenated to produce it.
Propionitrile is a chemical compound that is used in the pharmaceutical industry to create active pharmaceutical ingredients (APIs), such as analgesics, antiepileptic medications, and antihypertensive medicines. In the agrochemical sector, it is also utilised in the synthesis of fungicides, insecticides, and herbicides.
Despite being widely used, propionitrile has certain health risks and needs to be treated carefully. It is a poisonous and combustible material that can irritate the skin, eyes, and respiratory system, and in severe circumstances, result in death. Therefore, when handling propionitrile, appropriate safety procedures and precautions should be performed.





Sign in to leave a comment.