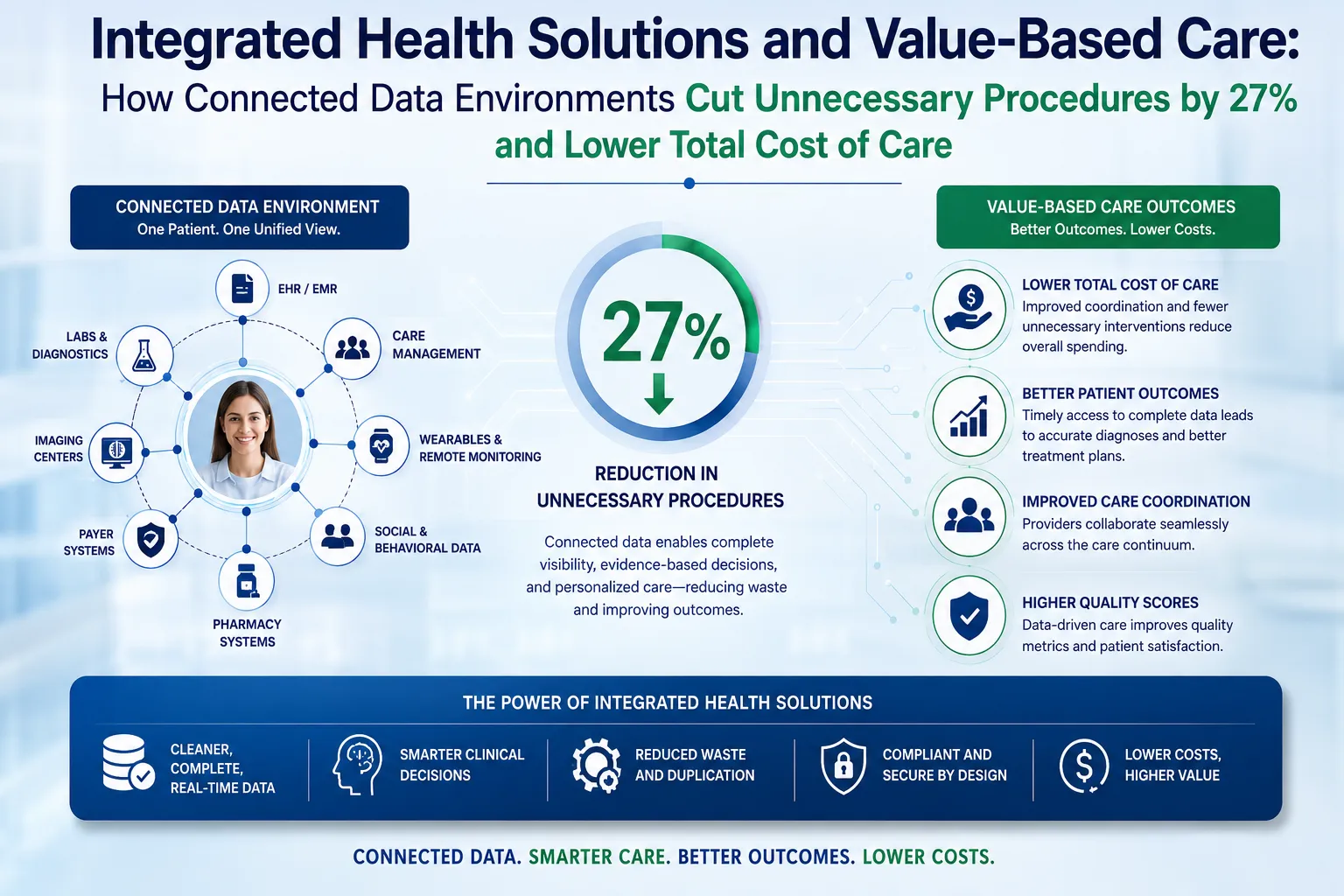
The year 2026 marks a turning point in modern medicine. Artificial intelligence, once the preserve of research labs and tech campuses, has now embedded itself into clinical workflows, diagnostic pipelines, and patient care pathways across the globe. But perhaps nowhere is its impact more profound than at the intersection of AI in healthcare and genomics — specifically, the rapidly evolving world of DNA testing.
From early disease detection to personalised treatment protocols, AI-powered DNA testing for diseases is fundamentally changing what it means to receive a diagnosis. At the same time, tools that started as consumer novelties — ancestry DNA testing platforms — are being repurposed to contribute to population-level health intelligence.
This article explores the state of play in 2026: where AI and DNA testing intersect, what it means for patients and healthcare professionals, and why this convergence represents one of the most significant shifts in preventive and precision medicine in a generation.
The AI Revolution in Healthcare: A 2026 Snapshot
AI in healthcare has moved well beyond proof-of-concept. According to the World Health Organisation's 2025 Digital Health Progress Report, over 60% of high-income countries have integrated at least one AI-assisted diagnostic tool into their national health infrastructure. In clinical settings, AI is being used to read radiology scans, flag anomalies in blood panels, predict patient deterioration in ICUs, and — increasingly — interpret complex genomic data at scale.
Three factors have driven this acceleration:
- Exponential growth in health data availability, including electronic health records, wearable biosensor outputs, and genomic datasets
- Advances in large-scale machine learning models capable of identifying patterns across millions of data points simultaneously
- Regulatory frameworks in the EU, US (via the FDA's AI/ML Action Plan), and UK (via MHRA guidance) that now provide clearer pathways for AI-powered medical devices
The result is a healthcare ecosystem in which AI does not replace clinicians but augments their capacity to diagnose earlier, more accurately, and more cost-effectively.
Key stat: The global AI in healthcare market is projected to reach $188 billion USD by 2030, growing at a CAGR of over 37% from 2024 — driven significantly by genomic AI applications. (Grand View Research, 2025)
DNA Testing in 2026: Far Beyond the Family Tree
Consumer DNA testing first captured the public imagination through ancestry DNA testing services — platforms that could map a person's ethnic heritage, identify distant relatives, and trace migration patterns across centuries. What began as a genealogy tool has since evolved into something far more clinically significant.
In 2026, ancestry DNA testing databases — with the informed consent of participants — are providing researchers and public health bodies with unprecedented genomic diversity data.
This matters enormously for AI training. Historically, genomic research was skewed toward populations of European descent. Today, datasets drawn from commercial ancestry platforms are helping to correct that imbalance, making AI diagnostic models more equitable and generalizable across ethnicities.
But the real clinical frontier lies in targeted DNA testing for diseases. This encompasses:
- Carrier screening: identifying whether individuals carry gene variants associated with hereditary conditions such as cystic fibrosis, sickle cell disease, or Fragile X syndrome
- Predictive genomic testing: assessing polygenic risk scores (PRS) for conditions like Type 2 diabetes, cardiovascular disease, and certain cancers
- Pharmacogenomic testing: determining how a patient's genetic makeup affects their response to specific medications — enabling precision prescribing and reducing adverse drug reactions
- Somatic tumour profiling: A medical research technique used to analyze a person’s DNA of cancer cells and relate it to any targetable mutations in their lifetime. This helps guide oncologists to take a treatment decision.
AI is the engine making all of this clinically actionable. Whole genome sequencing generates vast quantities of raw data — too much for any human analyst to interpret efficiently. AI models trained on linked genomic and clinical datasets can identify which variants are clinically meaningful, prioritise findings for clinical review, and generate structured reports that integrate directly into electronic health records.
How AI Is Transforming DNA Testing for Diseases?
The clinical integration of AI and DNA testing for diseases represents a step change in early detection medicine. Here are four areas where the impact is most pronounced in 2026:
1. Polygenic Risk Scoring at Population Scale
Polygenic risk scores aggregate the effect of thousands of common genetic variants to estimate an individual's predisposition to complex conditions. AI models can now generate and interpret PRS for cardiovascular disease, breast cancer, prostate cancer, and Alzheimer's disease with a level of accuracy that was not achievable five years ago.
Several NHS integrated care boards in England have piloted AI-powered PRS screening programmes through primary care, flagging high-risk patients for earlier intervention. Early results from the Genomics England pilot (2024-2025) showed a 28% improvement in early-stage cancer detection rates among genomically screened cohorts versus standard care.
Clinical note: Polygenic risk scores do not diagnose disease — they estimate relative risk. Patients receiving high-risk PRS results should always be guided by a qualified clinician or genetic counsellor to contextualise findings within their broader clinical picture.
2. AI-Powered Rare Disease Diagnosis
Rare diseases affect approximately 300 million people globally, yet the average diagnostic journey spans 4 to 7 years. Whole exome and whole genome sequencing, combined with AI phenotype-matching tools, are dramatically compressing this timeline.
Platforms such as Genix.ai are now capable of analyzing a patient's genomic sequence, using AI Technology, against databases of known disease variants and clinical phenotype data in hours rather than weeks. In pediatric rare disease settings, this speed can be the difference between effective early intervention and irreversible developmental harm.
3. Oncology: DNA Testing Meets Real-Time AI
Cancer genomics has become one of the most dynamic intersections of DNA testing and AI in healthcare. Liquid biopsy technology — which detects tumour-derived circulating DNA (ctDNA) from a blood sample — combined with AI interpretation, enables clinicians to:
- Monitor treatment response in real time without invasive tissue biopsies
- Detect minimal residual disease following surgery or chemotherapy
- Identify emerging resistance mutations before clinical relapse becomes apparent
Many companies have integrated AI interpretation layers into their liquid biopsy platforms, with several products now holding CE marking in Asia, Europe, and in North America.
4. Pharmacogenomics: Precision Prescribing
One of the most immediately applicable uses of DNA testing for diseases is pharmacogenomics — matching drug prescriptions to a patient's genetic profile. Variants in genes such as CYP2D6, CYP2C19, and TPMT determine how efficiently patients metabolize specific treatments, including antidepressants, antiplatelet agents, and chemotherapy drugs.
In 2026, AI-enabled pharmacogenomics decision support tools are being deployed directly into prescribing software, flagging gene-drug interactions at the point of care. The Dutch Pharmacogenomics Working Group (DPWG) guidelines, now embedded in multiple EHR systems across Europe, are increasingly powered by AI interpretation engines that contextualise variant data against current medication lists.
The Role of Ancestry DNA Testing in Population Health
It would be a mistake to view ancestry DNA testing as separate from the clinical genomics landscape. In 2026, the data infrastructure built by consumer genomics companies has become a meaningful contributor to public health intelligence.
23andMe (post-restructuring under Regeneron Pharmaceuticals), AncestryDNA, and MyHeritage DNA have all established research partnerships with academic medical centres and pharmaceutical companies. With participant consent, de-identified genomic data from ancestry databases is being used to:
- Identify novel disease-associated genetic variants across diverse populations
- Power genome-wide association studies (GWAS) at a scale previously impossible outside national biobanks
- Improve the diversity representation in AI training datasets, addressing longstanding biases in genomic research
For individuals, ancestry DNA testing increasingly comes bundled with optional health reports — polygenic risk score outputs, carrier status flags, and wellness trait analyses — all interpreted through AI algorithms. Consumers are advised to treat these as indicative rather than diagnostic, and to discuss any significant findings with their GP or a certified genetic counsellor.
Ethical Considerations: Genomic Data, Privacy, and Equity
No discussion of AI in healthcare and DNA testing is complete without addressing the ethical dimensions. Three areas warrant particular attention in 2026:
Data privacy and consent: Genomic data is uniquely identifying. Unlike a password, it cannot be changed. The General Data Protection Regulation (GDPR) in Europe classifies genetic data as a special category requiring explicit consent. Healthcare providers and testing companies must implement robust data governance frameworks and be transparent with patients about how their genomic information is stored, shared, and used.
Algorithmic bias: AI models trained predominantly on genomic data from populations of European ancestry may underperform when applied to patients of other ethnic backgrounds. This is an active area of concern and research — the goal of equitable AI in genomics requires diverse, representative training datasets.
Access and health equity: Advanced genomic testing and AI diagnostics carry costs. Without deliberate policy intervention, the benefits of precision genomics risk becoming concentrated among wealthier populations and health systems, widening existing health inequalities.
Looking Ahead: The Future of AI and DNA Testing in Healthcare
The convergence of AI in healthcare and DNA testing is not a future possibility — it is a present reality. In 2026, these technologies are saving lives through earlier cancer detection, accelerating rare disease diagnosis, enabling more precise prescribing, and beginning to bring population-scale genomic intelligence to bear on public health strategy.
Ancestry DNA testing, once a tool for tracing family histories, is now a contributor to the broader genomic data ecosystem powering AI research. And DNA testing for diseases, supported by AI interpretation, is moving from specialist genetics clinics into mainstream primary and secondary care.
For healthcare professionals, staying abreast of these developments is no longer optional — it is a clinical imperative. For patients, understanding the potential and the limitations of genomic testing has never been more important. And for the healthcare system as a whole, the challenge is to ensure that the extraordinary promise of AI-powered genomic medicine is delivered equitably, ethically, and in the genuine service of patient outcomes.













Sign in to leave a comment.