
Healthcare Quality Management Software helps organizations automate compliance processes, streamline audits, manage documents, track CAPA, and improve regulatory readiness. Qualityze simplifies healthcare quality management through centralized workflows, real-time visibility, and automated compliance solutions that improve efficiency, reduce risks, and support continuous quality improvement.

Environmental, Health, and Safety (EHS) management is undergoing a significant transformation with the integration of Artificial Intelligence (AI). As organi...
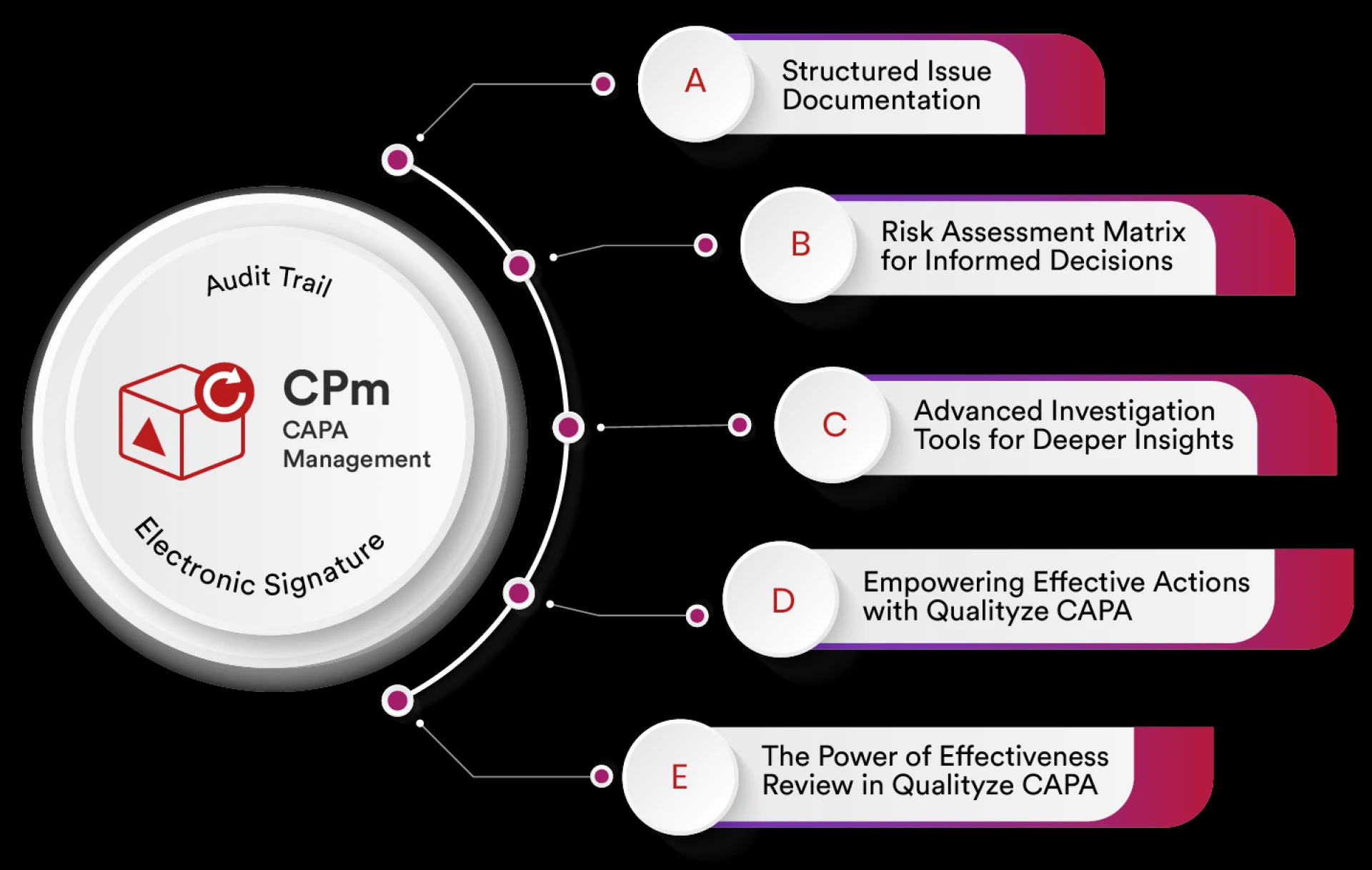
Corrective and Preventive Action (CAPA) is a core element of an effective Quality Management System. Organizations across pharmaceuticals, medical dev
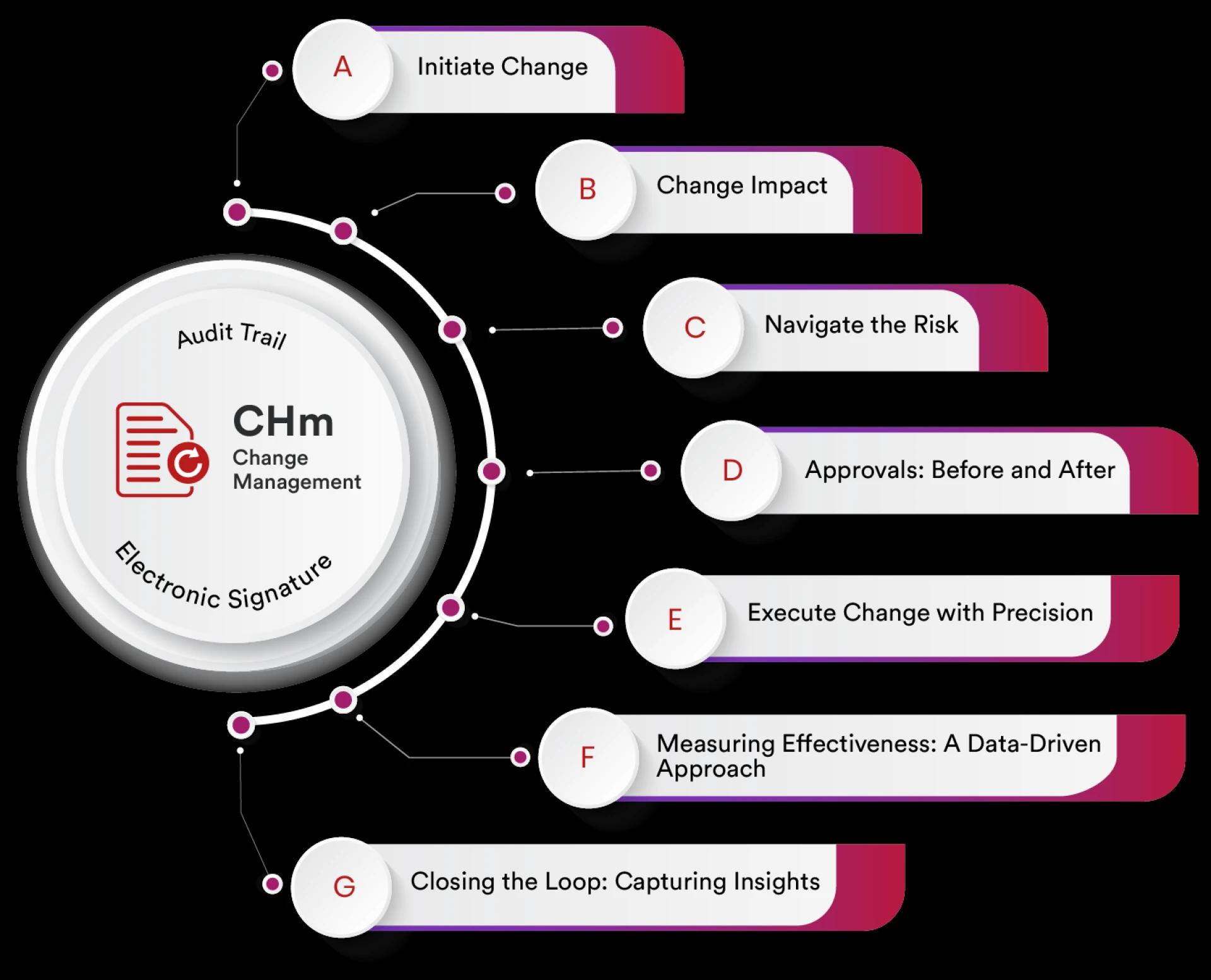
In today’s competitive business landscape, managing organizational change effectively is critical to maintaining quality, compliance, and operationa
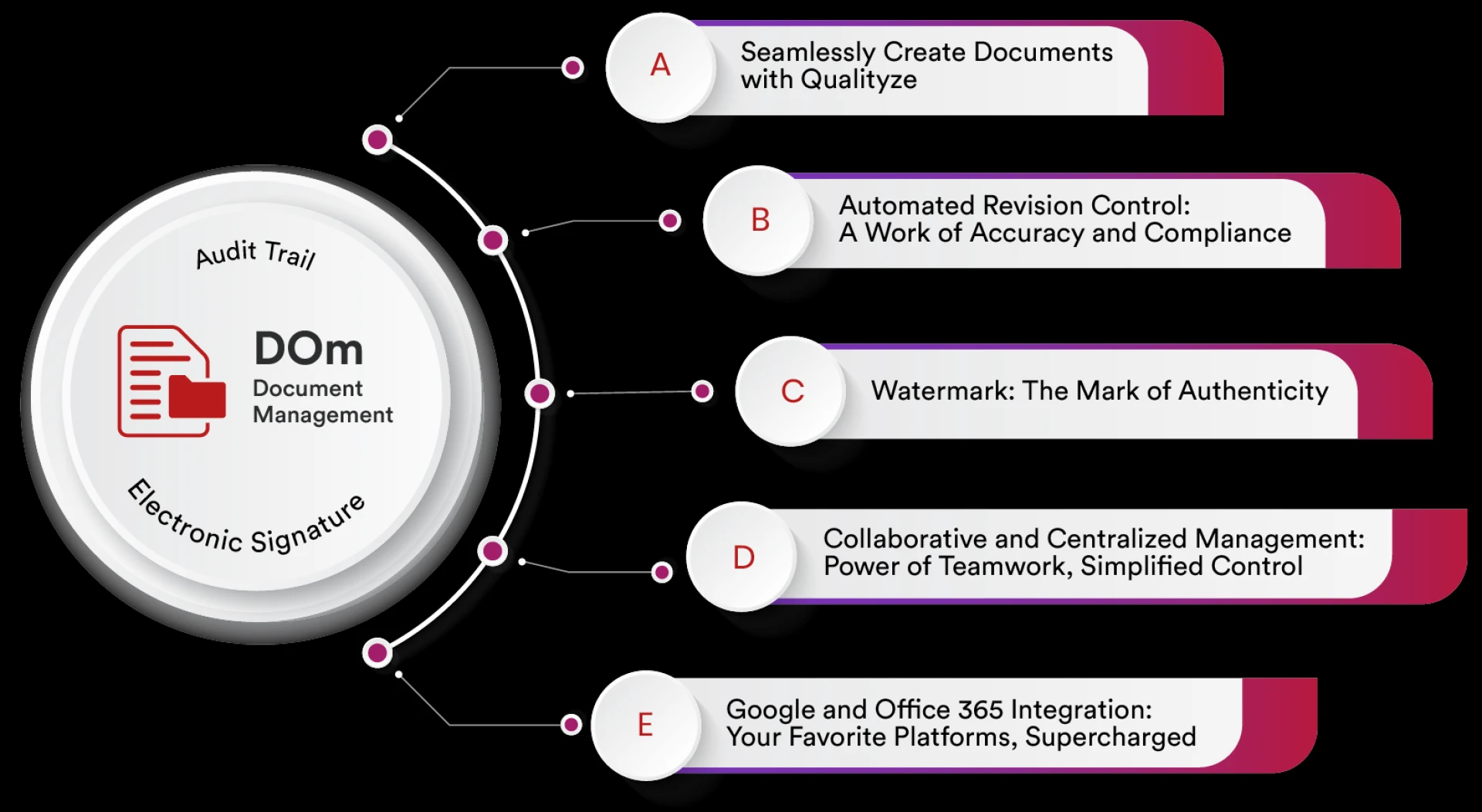
Enterprises in the USA face increasing challenges in managing vast volumes of documents, ensuring compliance, and maintaining operational efficiency.
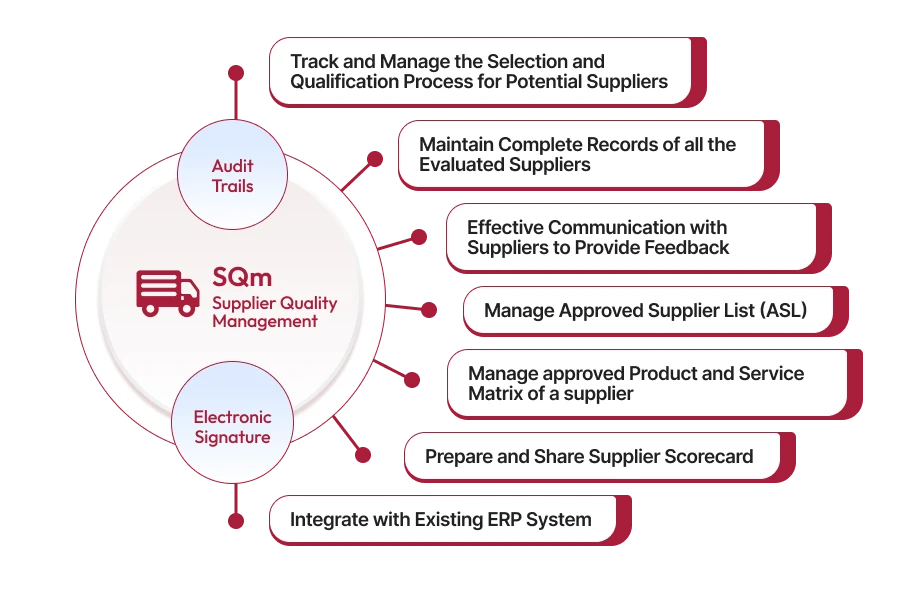
IntroductionIn today’s interconnected global supply chains, ensuring product quality starts long before manufacturing—it begins with your supplier
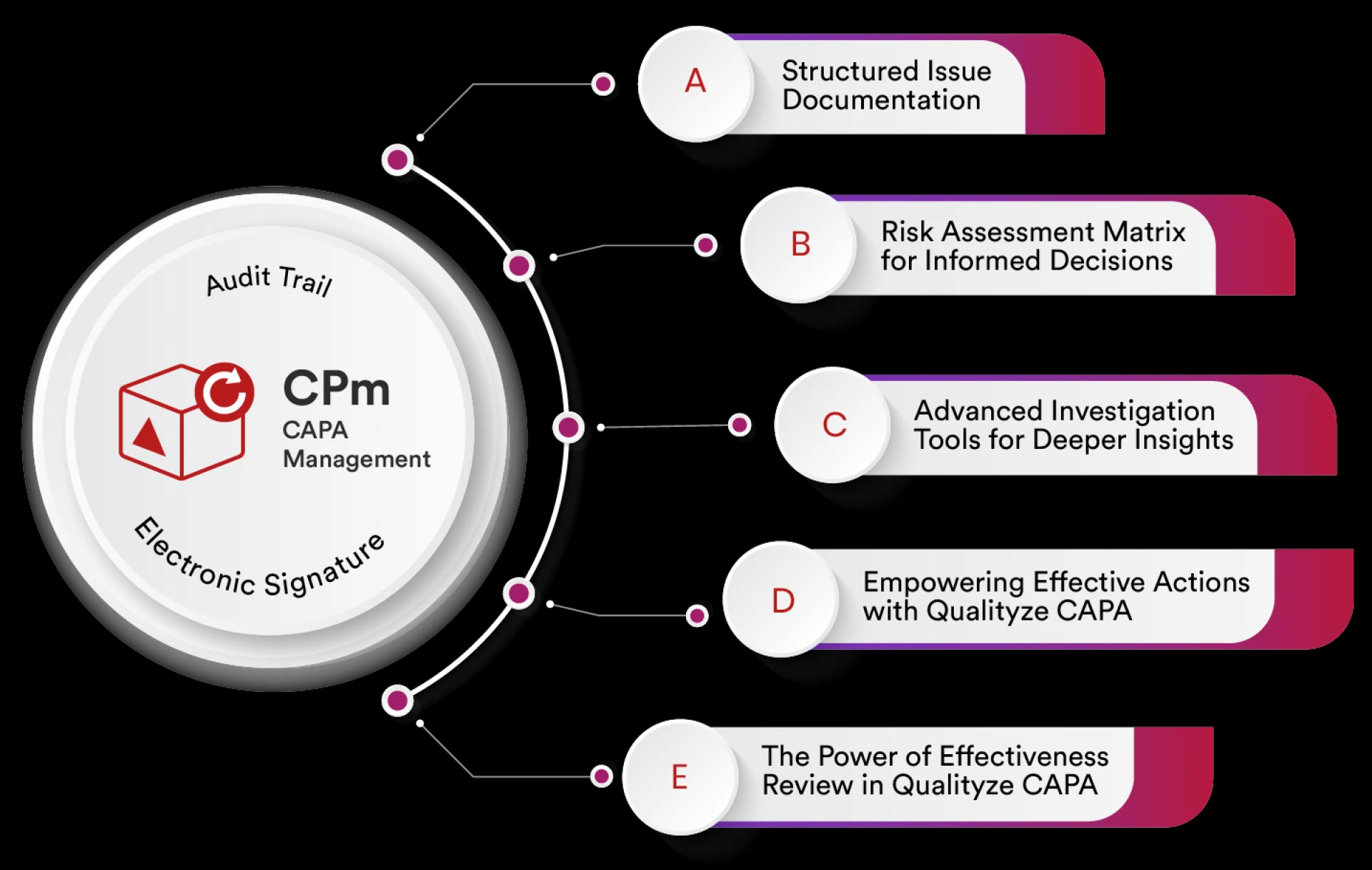
Qualityze offers a CAPA (Corrective and Preventive Action) software solution that integrates robust risk management features, making it particularly s

Qualityze Document Management simplifies management of growing volume of documents by providing a robust framework
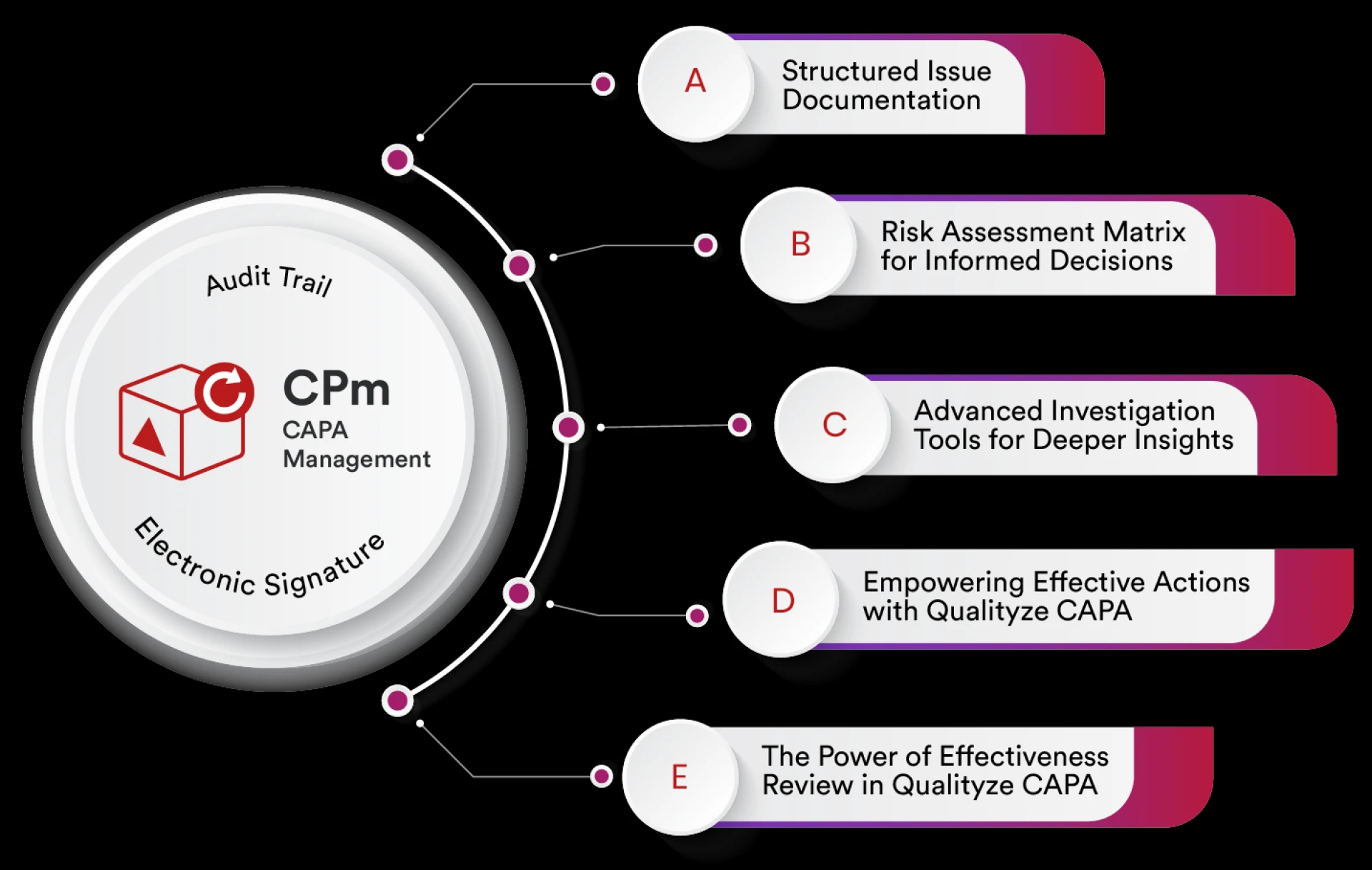
Whether you want to limit the reoccurrences of the same problems in your products or processes over and over again or want to improve the quality of y



