



Global pharmaceutical company Lupin Limited has announced the launch of Eslicarbazepine Acetate Tablets in the United States following approval from the U.S....

Lupin Limited has announced the launch of Risperidone for Extended-Release Injectable Suspension in the United States, strengthening its presence in t

Lupin Limited (Lupin), a leading global pharmaceutical organization, today announced that it has secured an S&P Global ESG Score of 91 for 2025, significantly higher than the pharmaceutical industry average of 28.

Mumbai, November 12, 2025: Lupin Limited (Lupin), a global pharmaceutical major, announced today that it has received the Establishment Inspection Rep

Mumbai, November 8, 2025: Lupin Limited, a global pharmaceutical major, reported that the U.S. Food and Drug Administration (U.S. FDA) has concluded a

Mumbai, November 5, 2025: Lupin Limited, a globally respected pharmaceutical company, announced today that it has received an Establishment Inspection

Mumbai, Naples, February 3, 2025: Lupin Limited (Lupin), a leading global pharmaceutical company, announced that it has received tentative approval fr
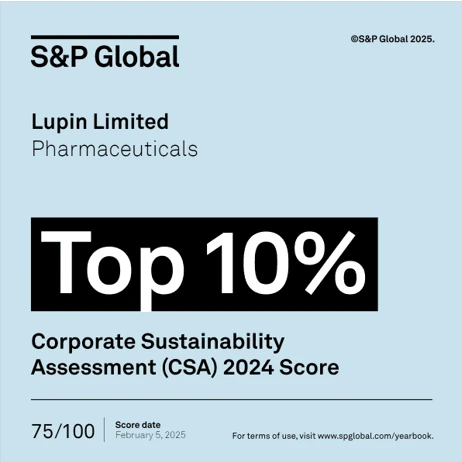
Mumbai, February 27, 2025: For the second consecutive year, Lupin Limited (Lupin) has earned a coveted place in the S&P Global Sustainability Year

Mumbai, February 19, 2025: Pharmaceutical leader Lupin Limited has been awarded an “A-” leadership rating by CDP for its performance in climate ch

Mumbai, February 21, 2025: Pharmaceutical major Lupin Limited has confirmed the receipt of an Establishment Inspection Report (EIR) from the U.S. FDA

Global pharmaceutical company Lupin Limited today confirmed the launch of Rivaroxaban Tablets USP, 2.5 mg, in the U.S., post the final FDA approval of