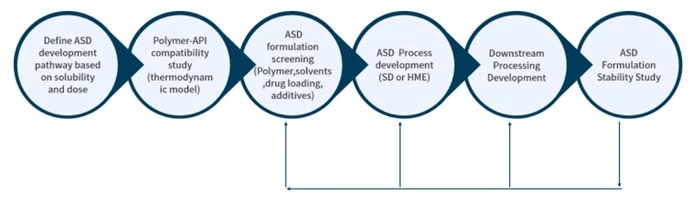
Crystal Pharmatech, the leading solid state research CRO, is proud to announce that the formulation platform of its subsidiary candoo packaging Company Inc. to address solubility and bioavailability issues has been featured in Drug Development and Delivery Journal in March 2023, titled as "SPECIAL FEATURE – Solubility & Bioavailability: Difficult Beasts to Tame"and can be accessed online.
Candoo Pharmatech Company Inc. is making strides in improving bioavailability and solubility for molecules to be transformed into medicines. It's an honor to be featured alongside renowned industry leaders such as Catalent, Evonik, Quotient and we're excited to contribute to this critical conversation.
The publication of our technology in this highly respected journal is a significant accomplishment as it demonstrates our commitment to advancing the field of pharmaceuticals and is a testament to the innovation and hard work of our team. As a team, we're committed to pushing the boundaries of pharmaceutical development and providing cutting-edge solutions that improve the lives of patients around the world.
As a professional cdmo pharma company, crystal biotech trys our best to provide good products and service for you.
For more information about contract research pharma, please feel free to contact us!
















Sign in to leave a comment.