India’s stent industry has grown from import dependence to global supply. Over the last decade, Indian companies have moved from producing basic bare-metal stents to advanced drug-eluting stents. With a strong push from domestic policies and growing demand abroad, these firms are now competing in the global stent market.
From Import Reliance to Local Strength
In the early 2000s, India relied heavily on imported stents. These products were expensive and often out of reach for the average patient. The local industry responded with low-cost, high-quality alternatives. This shift was supported by national programs like Make in India, which encouraged homegrown manufacturing in healthcare.
Today, Indian companies produce a wide range of coronary stents. Many of these are CDSCO-approved stents, meeting safety and quality benchmarks set by India’s drug regulator. Indian firms are also meeting international norms like ISO 13485, CE marking, and even USFDA approvals. This has helped open up overseas markets.
Innovation in Indian Stent Manufacturing
Innovation has been key to India’s rise in this space. Manufacturers have invested in in-house research to design better drug coatings, thinner struts, and delivery systems that work well in complex procedures. Bioabsorbable polymers and dual-drug technologies are also part of this development.
Some firms are using data tools to track stent performance over time. Others are testing new materials to improve flexibility and reduce restenosis. This innovation in Indian stent manufacturing is not only improving outcomes but also keeping costs down.
Indian stent developers often run their own clinical trials. This gives them more control over product testing and speeds up the path to market. These trials also build trust with international buyers and regulators.
Regulatory Advantage and Speed
The Central Drugs Standard Control Organization (CDSCO) has helped fast-track approvals for well-researched products. The regulator encourages trials done in India and offers clarity in approval timelines. CDSCO-approved stents are now seen as reliable and safe, which supports both domestic use and exports.
Regulatory familiarity has also made Indian firms more agile. They understand the complex paperwork needed for global approvals and adapt their systems quickly. This speed helps them enter new markets faster than many large global players.
Growth in Global Markets
India exports stents to more than 100 countries. Key markets include Southeast Asia, Latin America, the Middle East, and parts of Africa. Many Indian products have CE certification, allowing entry into select European countries as well.
Exports are growing in both value and volume. Indian stents are priced lower than Western brands, but quality is often at par. This makes them a good choice for countries where affordability is important.
Some companies also work under OEM contracts, producing for foreign brands. Others sell under their own label through local distributors or direct hospital partnerships.
India’s price cap policy has made companies more cost-focused. They design products that perform well but don’t add expensive, unneeded features. This approach has found acceptance in global markets looking for cost-effective care.
Focus on Manufacturing Scale and Support
Indian firms are building larger facilities and expanding automation. Some units are part of government-backed medical device parks. These clusters offer shared infrastructure, quality testing labs, and faster logistics.
Companies are also strengthening their support services. They train clinicians, offer product demos, and provide after-sales support in multiple countries. This helps them compete not just on product quality but also on service.
Post-market surveillance is another growing area. Indian firms are using global feedback to improve stent design and durability. This feedback loop is critical to building long-term credibility in export markets.
Future Outlook
Demand for stents is expected to rise worldwide. Aging populations, lifestyle changes, and wider cardiac screening are key drivers. The global stent market is looking for reliable suppliers who can scale and innovate at the same time.
India’s strength lies in its ability to offer made-in-India stents that meet global needs without high costs. With support from policies like the Production-Linked Incentive (PLI) scheme and a strong clinical base, the country is well-positioned to expand its share in this market.
Next-gen stents using biodegradable materials, smart coatings, and improved delivery systems are under development. Some Indian firms are also exploring AI-based tracking tools and remote monitoring integration.
Education and trust-building will remain important. Companies are working on physician outreach and better documentation to meet global procurement standards.
Conclusion
Stent manufacturers in India have moved from domestic suppliers to global players. Their products are present in over 100 countries and are gaining trust across markets. With smart design, proven safety, and price control, Indian firms are ready to compete in the growing global stent market.
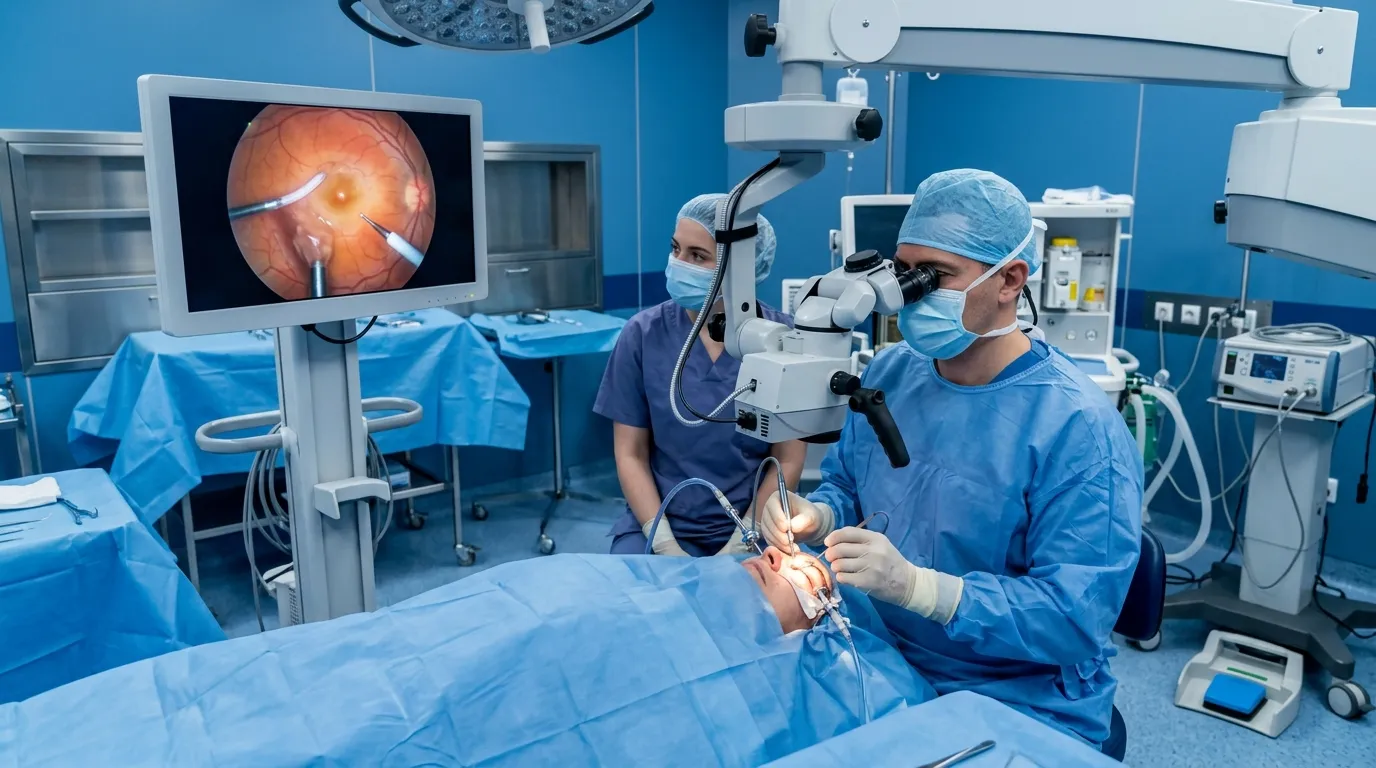
One innovation in this space comes from Translumina, which launched the world’s first balloon catheter with Trac+ patented premoulded tip technology. This design improves navigation through complex arteries, reduces vessel injury, and simplifies procedures. As demand for safer and more accurate treatment grows, such advances will continue to shape cardiovascular care globally.













Sign in to leave a comment.