

Navigating FDA Certification for Medical Devices
As a medical device manufacturer, getting your product FDA-certified isn’t just a legal requirement—it’s a crucial step toward ensuring yo

Similar Reads
Browse topics →
Why Consult an Experienced Endodontist in Nagpur for Complex Root Canal …

Recalibrating the Brain's 'Check Engine' Light: A Neurosurgeon's Take on…

The Brain's Weekend Pass: A Neurosurgeon on Why Vidalista 20's Greatest …

Advanced Techniques for Painless Wisdom Tooth Removal in Hinjewadi

The Garden of Your Second Brain: A Neurosurgeon on Amoxil's Unintended C…

The Brain's Surprise Party: A Neurosurgeon on Kamagra Jelly's Rapid Arri…

Freestanding vs. Built-In Baths: Which One Should You Choose for Your Ho…

Medicare Supplement Plans for 2026: What’s Changing, Who Should Enroll &…

Air Ambulance in Himachal Pradesh – Fast Medical Transport in Hills
More in Medicine & Healthcare
Browse all in Medicine & Healthcare → Medicine & Healthcare
Medicine & Healthcare
The Role of Caregivers in Managing Prescription Assistance for Aging Pat…
 Medicine & Healthcare
Medicine & Healthcare
Rethinking Waste and Access in the Diabetic Supply Ecosystem
 Medicine & Healthcare
Medicine & Healthcare
What Is In-Home Physical Therapy in Jupiter, FL?
 Medicine & Healthcare
Medicine & Healthcare
The Comprehensive Guide to Two-Wheeler Insurance
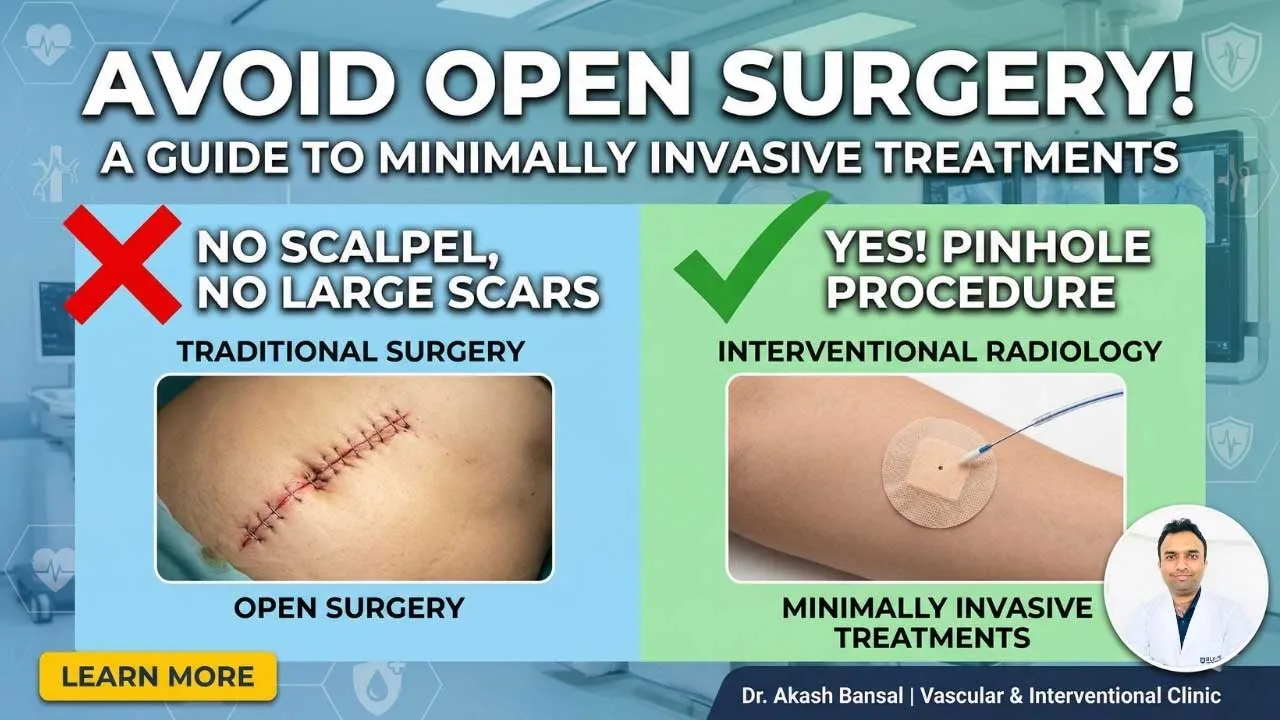 Medicine & Healthcare
Medicine & Healthcare
Avoiding Open Surgery: A Guide to Minimally Invasive Treatment
 Medicine & Healthcare
Medicine & Healthcare
Navigating the Complexities of Medical Billing in Illinois: A Guide to M…
Sign in to leave a comment.