Introduction
The clinical research services market rarely commands headlines, yet it quietly determines how quickly and how efficiently new therapies reach patients. At its core, the market reflects a structural shift in how pharmaceutical and biotechnology companies approach innovation. Drug development has become too complex, too expensive, and too globally dispersed to manage entirely in-house.
That reality has elevated clinical research service providers, especially contract research organizations (CROs), from peripheral vendors to strategic partners. What’s interesting is not just the growth of this market, but the subtle ways in which its role is evolving. It’s no longer about outsourcing tasks; it’s about outsourcing judgment, infrastructure, and increasingly, intelligence.
Market Overview: Scale Without Uniformity
Clinical research services represent the largest component of the broader CRO ecosystem, accounting for a significant share of outsourced R&D activity. In fact, clinical services alone contribute roughly three-quarters of total CRO service demand, underscoring their centrality in the value chain.
The volume of clinical trials continues to expand globally, with hundreds of thousands of studies registered and tens of thousands actively recruiting participants each year. This steady pipeline reflects not just scientific progress but also the rising burden of chronic diseases, which continues to push sponsors toward accelerated development timelines.
Yet the market is far from uniform. Growth rates vary across service lines—data management and regulatory services are expanding faster than traditional trial execution, reflecting a shift toward more specialized, knowledge-intensive offerings.
There’s also a structural imbalance: while clinical-phase services dominate revenue, early-stage and preclinical outsourcing still lag, suggesting untapped potential but also lingering sponsor caution.
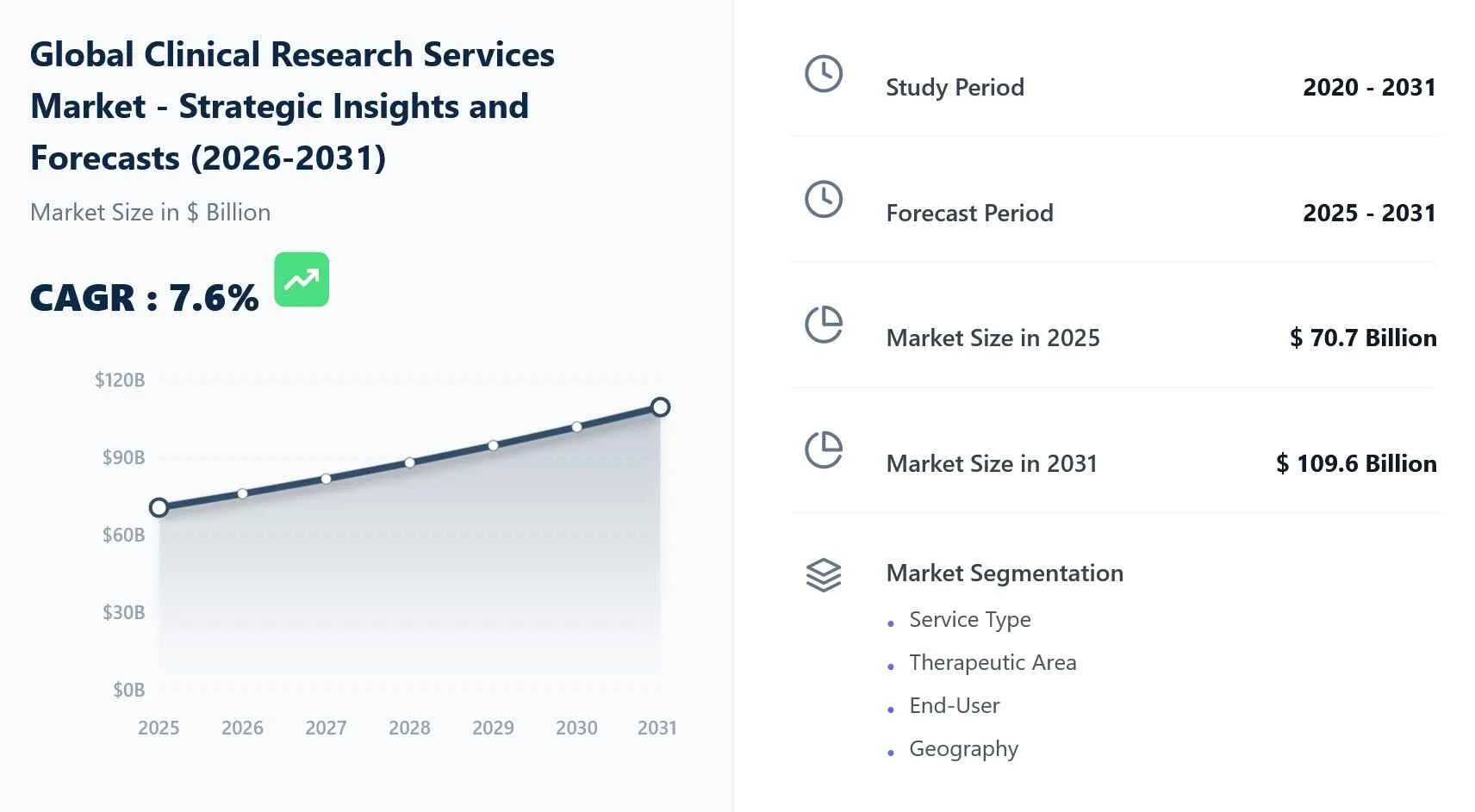
The global clinical research services market is projected to reach USD 109.6 billion by 2031, expanding at a steady CAGR of 7.6% from its 2025 valuation of USD 70.7 billion.
The Outsourcing Imperative: More Than Cost Savings
It’s tempting to frame outsourcing as a cost-cutting measure. That explanation is incomplete. Cost matters, but capability matters more.
Pharmaceutical companies now outsource more than half of their clinical trial activities, a sharp departure from the vertically integrated models of the past. The underlying reason is simple: internal teams struggle to keep pace with the complexity of modern trials.
Consider oncology studies, adaptive trial designs, or rare disease programs. These are not linear, predictable processes. They require specialized patient recruitment strategies, complex data architectures, and regulatory coordination across multiple jurisdictions. CROs, by design, aggregate this expertise.
In practice, outsourcing has become a way to de-risk execution rather than merely reduce expense. Sponsors are effectively trading control for predictability—and increasingly, for speed.
Complexity as a Growth Driver
Clinical trials are no longer standardized exercises. Protocols are more intricate, endpoints more nuanced, and patient populations more fragmented. This complexity is a primary driver of demand for clinical research services.
A significant proportion of new drug approvals now involve sophisticated trial designs, often spanning multiple geographies and therapeutic modalities. The operational burden of managing such studies internally can be overwhelming.
This is where CROs have found their edge. They offer pre-built infrastructure—site networks, data systems, regulatory pathways—that can be deployed quickly. Sponsors, in turn, gain flexibility without the need for long-term capital investment.
Interestingly, complexity doesn’t just increase demand; it reshapes the nature of services. Traditional monitoring and site management are giving way to integrated solutions that combine clinical operations, analytics, and compliance into a single offering.
Technology Integration: From Support Tool to Core Capability
If there is one theme redefining the clinical research services market, it is the integration of technology—not as an add-on, but as a core capability.
Artificial intelligence and machine learning are increasingly used to optimize trial design, identify suitable patient populations, and analyze complex datasets. These tools can reduce trial timelines and improve success rates, though their real value lies in decision support rather than automation.
Decentralized clinical trials (DCTs) represent another significant shift. By enabling remote patient monitoring and data collection, DCTs expand geographic reach and improve patient participation.
What’s notable is that technology adoption is uneven. Larger CROs tend to lead, leveraging scale to invest in proprietary platforms. Smaller players often struggle to keep up, which may accelerate consolidation over time.
There’s also a subtle tension: while technology promises efficiency, it challenges the traditional revenue model of CROs, which has historically been tied to labor-intensive services.
Data as a Strategic Asset
Clinical research has always been data-driven, but the nature of that data is changing. The volume is increasing, the sources are diversifying, and the expectations around its use are rising.
Data management is now one of the fastest-growing segments within clinical research services, driven by the need for accuracy, compliance, and real-time insights.
More importantly, data is becoming a competitive differentiator. CROs that can integrate clinical trial data with real-world evidence, genomics, and patient-reported outcomes are better positioned to offer value beyond execution.
This shift has strategic implications. It suggests that the future leaders in this market may look less like service providers and more like data companies with clinical capabilities.
Regulatory Complexity: A Double-Edged Sword
Regulation has always been a defining feature of clinical research, but its impact on the services market is nuanced.
On one hand, stringent regulatory requirements increase the need for specialized expertise, driving demand for CRO services. On the other, regulatory variability across regions introduces operational challenges and cost pressures.
Navigating ethics approvals, data privacy laws, and evolving compliance standards requires not just knowledge, but infrastructure. This again favors larger, globally integrated CROs.
At the same time, regulatory agencies are gradually adapting to new models, including decentralized trials and digital endpoints. The pace of this adaptation will influence how quickly the market evolves.
Competitive Landscape: Scale vs. Specialization
The clinical research services market is highly competitive, but not in a straightforward way.
Large, full-service CROs dominate in terms of revenue and global reach. They offer end-to-end solutions, which appeal to sponsors seeking simplicity and accountability. However, their size can be a disadvantage in certain contexts, particularly for smaller biotech firms that prefer more flexible, specialized partners.
Mid-sized and niche CROs often compete on expertise—focusing on specific therapeutic areas, trial phases, or technologies. This specialization can be a powerful differentiator, especially as trials become more complex.
That said, competition is intensifying. Providers are under pressure to innovate while managing costs, a balancing act that is not always sustainable.
The likely outcome is continued consolidation, alongside the emergence of hybrid models that combine scale with specialization.
Regional Dynamics: Growth Beyond Traditional Centers
North America remains the largest market for clinical research services, driven by strong pharmaceutical R&D activity and established infrastructure. However, growth is increasingly shifting toward Asia-Pacific.
Countries in this region offer a combination of cost advantages, large patient populations, and improving regulatory frameworks. For sponsors, this creates opportunities to accelerate recruitment and reduce trial costs.
At the same time, globalization introduces complexity. Managing multi-region trials requires coordination across diverse regulatory and operational environments—a challenge that again reinforces the value of experienced CRO partners.
Emerging Trends: Subtle but Significant Shifts
Several trends are shaping the future of the clinical research services market, though not all are immediately visible.
First, there is a move toward phase-specific outsourcing. Sponsors are increasingly selecting different providers for different stages of development, rather than relying on a single partner throughout.
Second, real-world evidence is gaining importance. Post-market data is no longer an afterthought; it is becoming integral to both regulatory approval and commercial strategy.
Third, patient-centricity is slowly gaining traction. While still more rhetoric than reality in some cases, there is a growing recognition that patient engagement can improve trial outcomes.
Finally, there is a gradual shift toward outcome-based partnerships, where CRO compensation is linked to performance metrics rather than time and materials. This model remains in its early stages but could redefine industry economics.
Challenges: Growth With Friction
Despite strong demand, the market faces several challenges.
Patient recruitment remains a persistent bottleneck. Delays in enrollment can significantly extend trial timelines and increase costs.
Talent shortages are another issue. Clinical research requires specialized skills, and competition for experienced professionals is intense.
There is also the question of data quality and integration. As trials become more complex, ensuring consistency and reliability across multiple data sources becomes increasingly difficult.
Finally, there is an inherent tension in the CRO business model. Efficiency improvements—driven by technology—can reduce billable hours, potentially impacting revenue. How providers adapt to this dynamic will be critical.
Outlook: Evolution Rather Than Disruption
The clinical research services market is not heading toward a sudden transformation. Instead, it is evolving gradually, shaped by incremental changes in technology, regulation, and sponsor behavior.
Growth is expected to remain steady, supported by rising R&D investment, increasing trial complexity, and continued outsourcing. At the same time, the nature of competition will shift—from scale to capability, from execution to insight.
Perhaps the most important change is conceptual. Clinical research services are no longer just about conducting trials. They are about enabling decisions—faster, better, and with greater confidence.
That shift may not be dramatic, but it is consequential. And it suggests that the future of this market will be defined less by how trials are run, and more by how knowledge is generated and applied.

















Sign in to leave a comment.