In today’s regulated life sciences, biotechnology, and MedTech industries, compliance isn’t optional—it’s essential. Regulatory bodies such as the FDA and EMA expect companies to demonstrate reliable systems, validated processes, and trustworthy data. The cost of non-compliance isn’t limited to failed audits; it can lead to reputational harm, delayed approvals, and even risks to patient safety.
At Compliance Gurus, we empower organizations to navigate complex compliance landscapes with expert guidance in data integrity services, computer software assurance (CSA), FDA frameworks, ALCOA+ principles, and Annex 11 software validation. This article explores these critical areas in detail and how our team helps companies achieve compliance success.
The Importance of Data Integrity Services
Data integrity is the foundation of compliance. Regulatory agencies define data integrity as maintaining the accuracy, completeness, and reliability of data throughout its lifecycle. Without robust data integrity systems, even the most advanced technology or processes can fail regulatory scrutiny.

Why Data Integrity Matters:
- Ensures data is accurate, consistent, and trustworthy
- Protects patient safety and clinical outcomes
- Meets FDA, EMA, and global regulatory requirements
- Builds confidence with auditors and stakeholders
Compliance Gurus provides data integrity services that include risk assessments, gap analyses, training programs, and remediation plans. Our tailored approach helps organizations strengthen governance frameworks and embed compliance into daily operations.
Computer Software Assurance: Modernizing Validation
Traditional Computer System Validation (CSV) has often been criticized as overly burdensome, with excessive documentation and limited focus on actual risk. Recognizing this, the FDA introduced Computer Software Assurance (CSA) as a smarter, risk-based framework.

Key Benefits of CSA:
- Focuses on critical quality and patient safety risks
- Reduces unnecessary documentation workload
- Accelerates software implementation timelines
- Aligns with FDA’s modernization vision
Compliance Gurus helps organizations adopt computer software assurance strategies that streamline validation processes. By emphasizing testing over paperwork, our approach saves time and resources while ensuring systems remain inspection-ready.
Understanding CSA FDA Guidance
The FDA’s CSA framework represents a significant shift in how software validation is approached. Instead of applying the same level of scrutiny to all systems, CSA promotes a proportional, risk-based approach.

What CSA FDA Means for Companies:
- Transitioning from documentation-heavy CSV to CSA
- Prioritizing risk-based testing strategies
- Leveraging automation tools for validation
- Meeting FDA expectations with greater efficiency
At Compliance Gurus, we guide organizations through this transition with structured roadmaps. Our CSA FDA services include training teams, re-designing validation frameworks, and implementing best practices to ensure ongoing compliance.
ALCOA+ Principles in Software Validation
Data must not only be captured but also maintained according to specific quality standards. The ALCOA+ principles—standing for Attributable, Legible, Contemporaneous, Original, and Accurate, plus Complete, Consistent, Enduring, and Available—are widely accepted as the gold standard for data integrity.
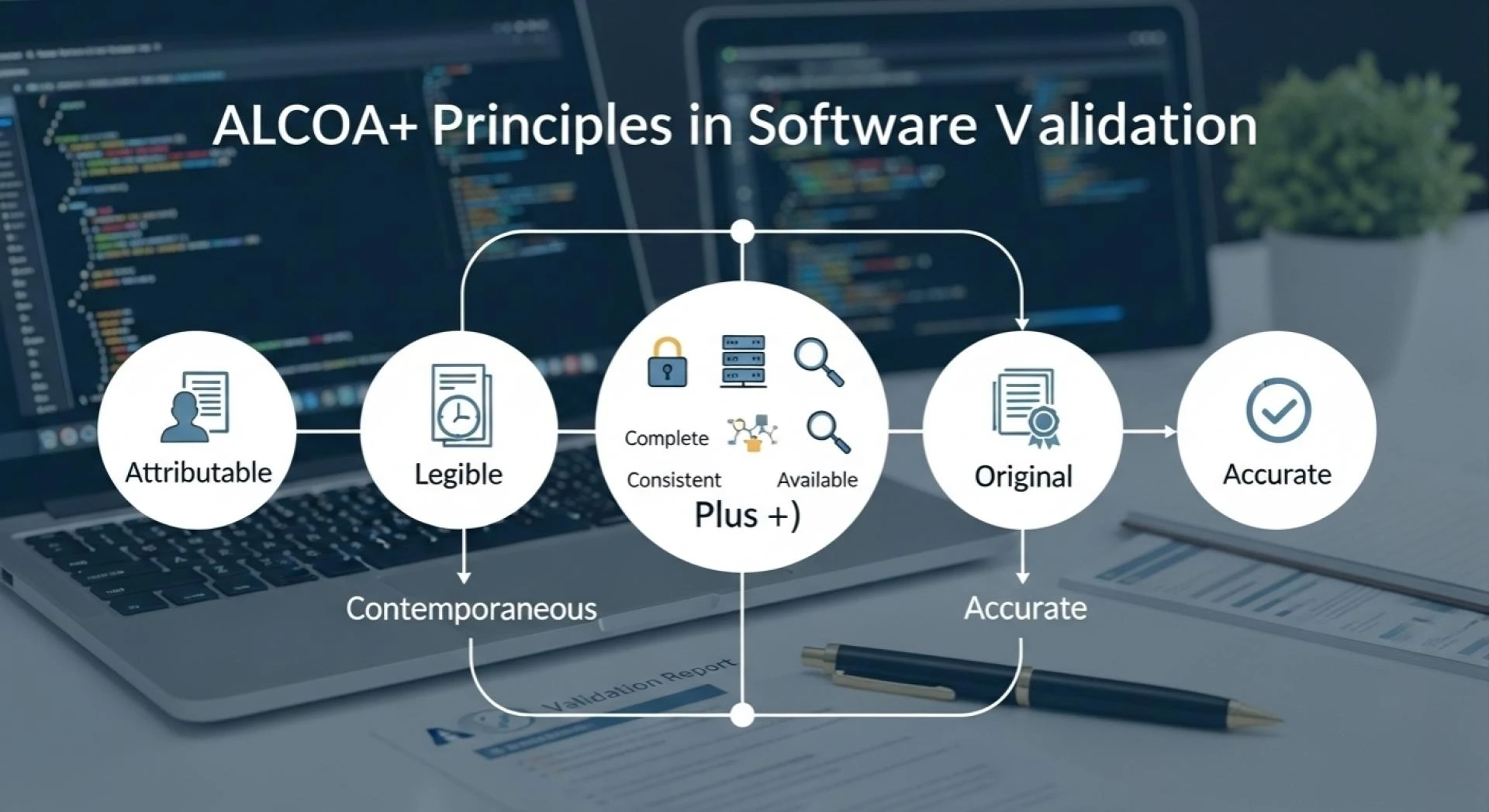
Applying ALCOA+ in Validation:
- Attributable: Every action must be linked to the individual performing it
- Legible: Records should remain clear and readable throughout the lifecycle
- Contemporaneous: Data must be recorded at the time of activity
- Original: Preserve original records or verified true copies
- Accurate: Records should be free from error and reflect reality
The “+” elements ensure completeness, consistency, durability, and accessibility. Compliance Gurus integrates ALCOA+ principles in software validation to ensure that validated systems protect data integrity at every stage.
Annex 11 Software Validation: EMA Perspective
While the FDA emphasizes CSA, the European Medicines Agency (EMA) enforces compliance through Annex 11 of the EU GMP guidelines. Annex 11 sets expectations for computerized systems used in regulated environments.

Core Requirements of Annex 11:
- Risk-based validation of computerized systems
- Clear roles and responsibilities for system ownership
- Audit trails for traceability
- Data protection and backup strategies
- Periodic reviews of system performance
Compliance Gurus provides Annex 11 software validation services to help organizations align with EMA requirements while maintaining global compliance consistency. Our experts design validation frameworks that address both FDA and EMA expectations, minimizing duplication of effort.
The Compliance Gurus Approach
At Compliance Gurus, we recognize that every organization has unique compliance challenges. Our methodology blends regulatory expertise with practical, risk-based strategies.
How We Deliver Value:
- Assessment & Gap Analysis: Identifying compliance gaps across systems and processes
- Tailored Strategies: Developing customized frameworks for data integrity, CSA, and Annex 11
- Implementation Support: Assisting with validation execution, documentation, and training
- Audit Readiness: Preparing teams and systems for successful FDA or EMA inspections
- Continuous Improvement: Embedding compliance culture for sustainable success
By partnering with us, organizations can transform compliance from a burden into a competitive advantage.
Why Choose Compliance Gurus?
- Expertise Across Regulations: Deep understanding of FDA, EMA, and global standards
- Risk-Based Methodology: Focused on efficiency and critical risks
- Tailored Services: Customized to your company’s needs and scale
- Audit-Ready Deliverables: Documentation and systems built to withstand regulatory scrutiny
- Trusted Partner: Proven track record in life sciences and MedTech compliance
Conclusion
In the rapidly evolving regulatory landscape, organizations must adapt to modern compliance expectations while ensuring patient safety and data reliability. Whether it’s data integrity services, computer software assurance, CSA FDA guidance, ALCOA+ principles, or Annex 11 software validation, Compliance Gurus provides the expertise to help companies stay ahead.
Compliance is more than a regulatory requirement—it’s a commitment to quality, safety, and trust. With Compliance Gurus as your partner, you can navigate complexities confidently and focus on what truly matters: delivering safe and effective solutions to patients worldwide.

















Sign in to leave a comment.