Neurostimulation treatments have revolutionized the way we manage chronic pain, neurological disorders, and even mental health conditions. From spinal cord stimulation (SCS) to deep brain stimulation (DBS) and transcranial magnetic stimulation (TMS), these techniques are often hailed as non-pharmacological alternatives for patients who have exhausted traditional therapies. However, as with any medical procedure, there are side effects—some minor, others potentially serious. This article offers a critical review of neurostimulation treatments, highlighting the potential side effects and considerations before opting for such interventions.
Introduction to Neurostimulation
Neurostimulation involves the application of electrical or magnetic impulses to modulate nervous system activity. It is commonly used to treat:
- Chronic pain
- Parkinson’s disease
- Epilepsy
- Depression
- Migraines
- Obsessive-compulsive disorder (OCD)
While clinical trials have shown promise, a review of neurostimulation treatments is incomplete without discussing the risks associated with these techniques.
Common Neurostimulation Treatments
1. Spinal Cord Stimulation (SCS)
Involves the implantation of electrodes near the spinal cord to block pain signals.
2. Deep Brain Stimulation (DBS)
Electrodes are implanted into specific areas of the brain and connected to a pulse generator.
3. Vagus Nerve Stimulation (VNS)
Targets the vagus nerve to treat epilepsy and depression.
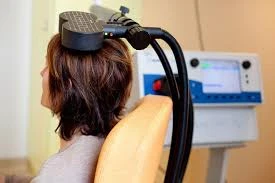
4. Transcranial Magnetic Stimulation (TMS)
A non-invasive method using magnetic fields to stimulate nerve cells in the brain.
Side Effects of Neurostimulation Treatments
1. Surgical Risks
For treatments like DBS and SCS, surgery is required. Common complications include:
- Infection
- Bleeding
- Hardware malfunction
- Anesthesia-related risks
This is a critical consideration in any review of neurostimulation treatments, especially for individuals with underlying health conditions.
2. Neurological Symptoms
DBS and other brain-targeted treatments can sometimes lead to:
- Headaches
- Speech issues
- Cognitive disturbances
- Seizures (in rare cases)
These side effects are generally transient but must be closely monitored.
3. Pain and Discomfort
Post-implantation pain, especially in the neck, back, or chest, can occur in patients undergoing SCS or VNS. Some users also report:
- Tingling sensations
- Muscle twitching
- Discomfort at the stimulation site
As noted in various reviews of neurostimulation treatments, such issues may diminish over time but are not uncommon in the initial stages.
4. Mood and Behavior Changes
Particularly with DBS and VNS, some patients report changes in mood, such as:
- Depression
- Anxiety
- Euphoria
- Irritability
This emphasizes the need for psychiatric evaluation both pre- and post-treatment.
5. Device-Related Complications
Breakage or movement of electrodes, battery failures, or improper functioning of the device can occur. Reprogramming or additional surgery might be required, making this an important concern in any comprehensive review of neurostimulation treatments.
6. Other Physical Reactions
These may include:
- Dizziness
- Nausea
- Skin irritation
- Swelling or bruising at implant sites
TMS, while non-invasive, can cause scalp discomfort or twitching of facial muscles during treatment.
Psychological and Cognitive Considerations
Research has shown that some neurostimulation treatments might influence cognitive functions. For instance:
- Memory problems have been reported in DBS patients.
- Altered decision-making or problem-solving skills
- Emotional blunting or overstimulation
Though rare, these side effects contribute to the ongoing debate and critical review of neurostimulation treatments.
Long-Term Safety and Efficacy
While most patients tolerate neurostimulation well, the long-term impact remains under investigation. Several factors influence long-term success:
- Individual medical history
- Treatment adherence
- Regular follow-ups for device calibration
The need for lifelong device management and possible future replacements (like battery changes) also adds to the complexity.
Ethical and Regulatory Concerns
Informed consent and patient autonomy are essential. Issues such as:
- Misdiagnosis before treatment
- Over-reliance on device-based solutions
- High costs and accessibility
...raise questions about the widespread use of neurostimulation, making this aspect integral to any review of neurostimulation treatments.
Who Should Avoid Neurostimulation?
Though many patients benefit from neurostimulation, certain groups should proceed with caution:
- Individuals with severe psychiatric disorders
- Those with active infections
- Pregnant women (especially in the case of TMS or VNS)
- Patients with cardiac pacemakers (device interference risk)
Pre-treatment screening is crucial for minimizing side effects and maximizing outcomes.
Patient Experiences and Case Studies
Various case studies and patient testimonials offer a mixed bag:
- Some report life-changing benefits, particularly in pain management or tremor control.
- Others experience minimal relief and substantial discomfort, leading to device removal.
Such divergent results underline the importance of personalized treatment planning and detailed reviews of neurostimulation treatments before making decisions.
Conclusion
Neurostimulation represents a promising frontier in modern medicine, offering hope for patients with treatment-resistant conditions. However, the potential side effects—ranging from mild discomfort to serious neurological or psychological disturbances—must not be overlooked. A comprehensive review of neurostimulation treatments is essential for clinicians and patients alike to make informed decisions.
Before opting for any neurostimulation therapy, thorough consultations, mental and physical health assessments, and long-term follow-up planning are imperative. As with any intervention, weighing benefits against risks is the key to safe and effective treatment.
FAQs on Neurostimulation Treatments
1. What are neurostimulation treatments used for?
They are used for conditions like chronic pain, epilepsy, depression, and Parkinson’s disease.
2. Are neurostimulation treatments safe?
Generally yes, but they carry risks like infection, mood changes, or hardware issues.
3. Can neurostimulation cause permanent damage?
Rarely. Most side effects are temporary, but there is always a small risk of complications.
4. How long does a neurostimulation device last?
Batteries may last 3-7 years and can be replaced surgically.
5. Is the treatment painful?
The surgery may cause temporary pain; stimulation itself is usually painless.
6. Are there any alternatives to neurostimulation?
Yes—medications, physical therapy, psychotherapy, and lifestyle changes.
7. Can I undergo MRI with an implanted device?
Some devices are MRI-compatible; consult your doctor before scheduling scans.
8. How quickly will I see results?
Some patients experience immediate relief; others may take weeks or months.
9. Is neurostimulation covered by insurance?
It depends on your location and insurance plan; partial coverage is common.
10. Who qualifies for neurostimulation therapy?
Patients with chronic, treatment-resistant conditions who pass screening evaluations.
11. Are neurostimulation devices visible?
Implants are generally discreet, though some bulging under the skin may occur.
12. Can neurostimulation help with anxiety?
Certain types like TMS may help with anxiety, though it's not universally approved.
13. What is the recovery time after device implantation?
Recovery can range from a few days to several weeks, depending on the procedure.
14. Can the device be removed?
Yes, neurostimulation devices can be surgically removed if needed.
15. What follow-up care is required?
Regular appointments for monitoring, device adjustment, and battery replacement.













Sign in to leave a comment.