Introduction
Pharmaceutical intermediates are the building blocks of life-saving medicines. They play a crucial yet often overlooked role in the drug manufacturing process. At Dolphin Pharmaceutical, a leading pharmaceutical drug manufacturer in India and trusted intermediates manufacturer in India, we recognize the significance of these compounds in ensuring the quality, safety, and efficacy of the final pharmaceutical product.
This blog explores the critical role pharmaceutical intermediates play in drug development, the various types, regulatory standards, and how Dolphin Pharmaceutical is setting benchmarks in delivering high-quality intermediates for global markets.

1. What Are Pharmaceutical Intermediates?
Pharmaceutical intermediates are chemical compounds used in the synthesis of active pharmaceutical ingredients (APIs). They are not the final product but are essential stages in the multi-step process of drug development.
These compounds ensure that each successive chemical reaction is performed accurately, eventually leading to a stable and effective API.
Without high-quality intermediates, the entire manufacturing process can be compromised, affecting the safety and therapeutic efficacy of the medication.
How Intermediates Differ from APIs (Active Pharmaceutical Ingredients)
While APIs are the biologically active components of a drug, intermediates are their precursors. The key difference is that APIs are the final output ready for formulation into dosage forms like tablets or injections, whereas intermediates are the earlier products formed during chemical synthesis.
Both require strict quality control, but intermediates are often subject to fewer regulatory constraints—yet their importance in the supply chain cannot be underestimated.
2. The Role of Intermediates in Pharmaceutical Manufacturing
Step-by-Step Contribution to Final Drug Formulation
Intermediates serve as crucial checkpoints in the multi-stage synthesis of APIs. Each intermediate must meet specific purity and structural standards before it moves to the next stage.
This ensures that the final API is of consistent and high quality, ultimately leading to safer and more effective medications.
Why Quality of Intermediates Impacts Drug Efficacy and Safety
Contaminated or substandard intermediates can lead to ineffective or unsafe APIs. That’s why reputable intermediates manufacturers in India, like Dolphin Pharmaceutical, focus on maintaining purity levels, controlling impurities, and ensuring chemical stability throughout the process.
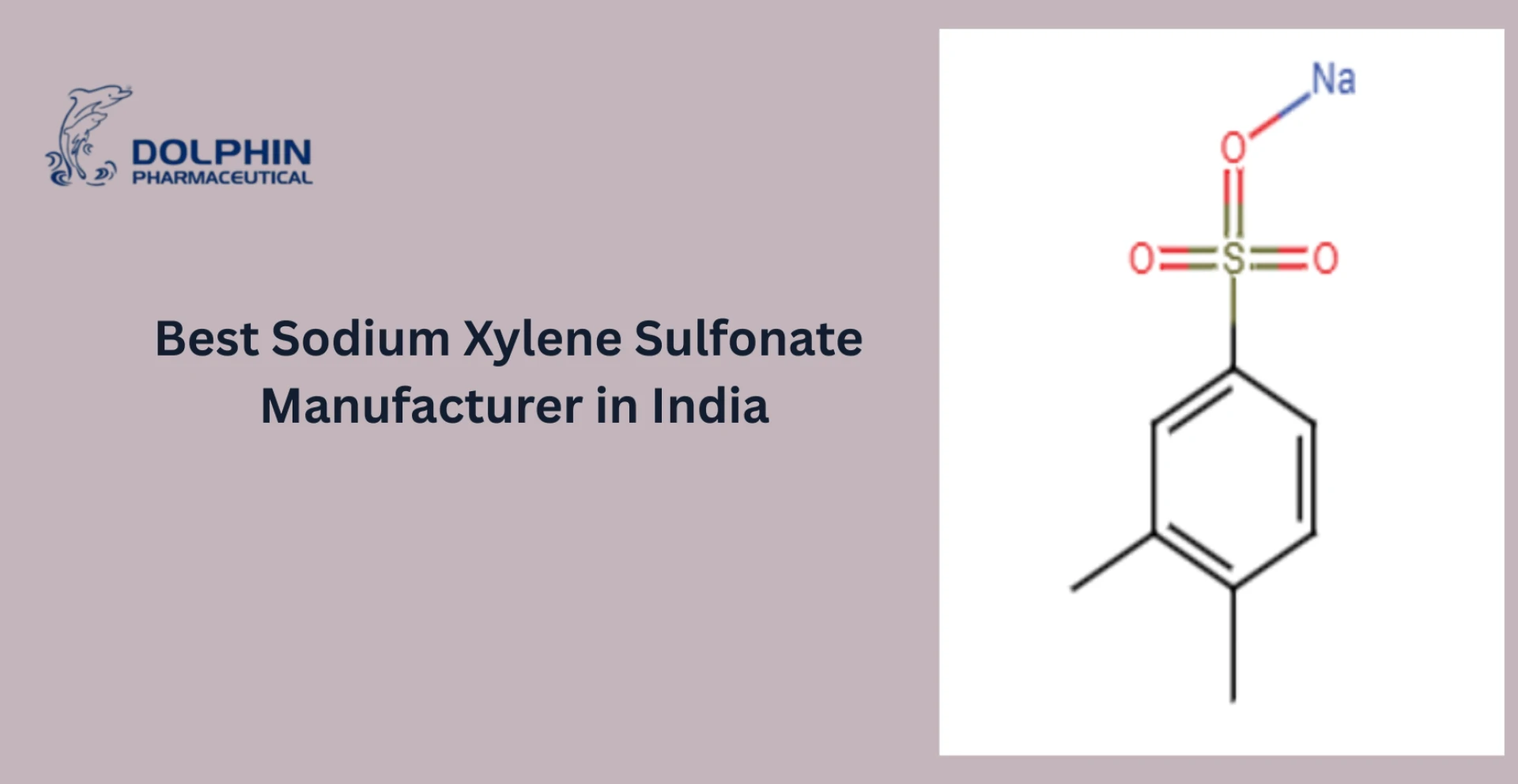
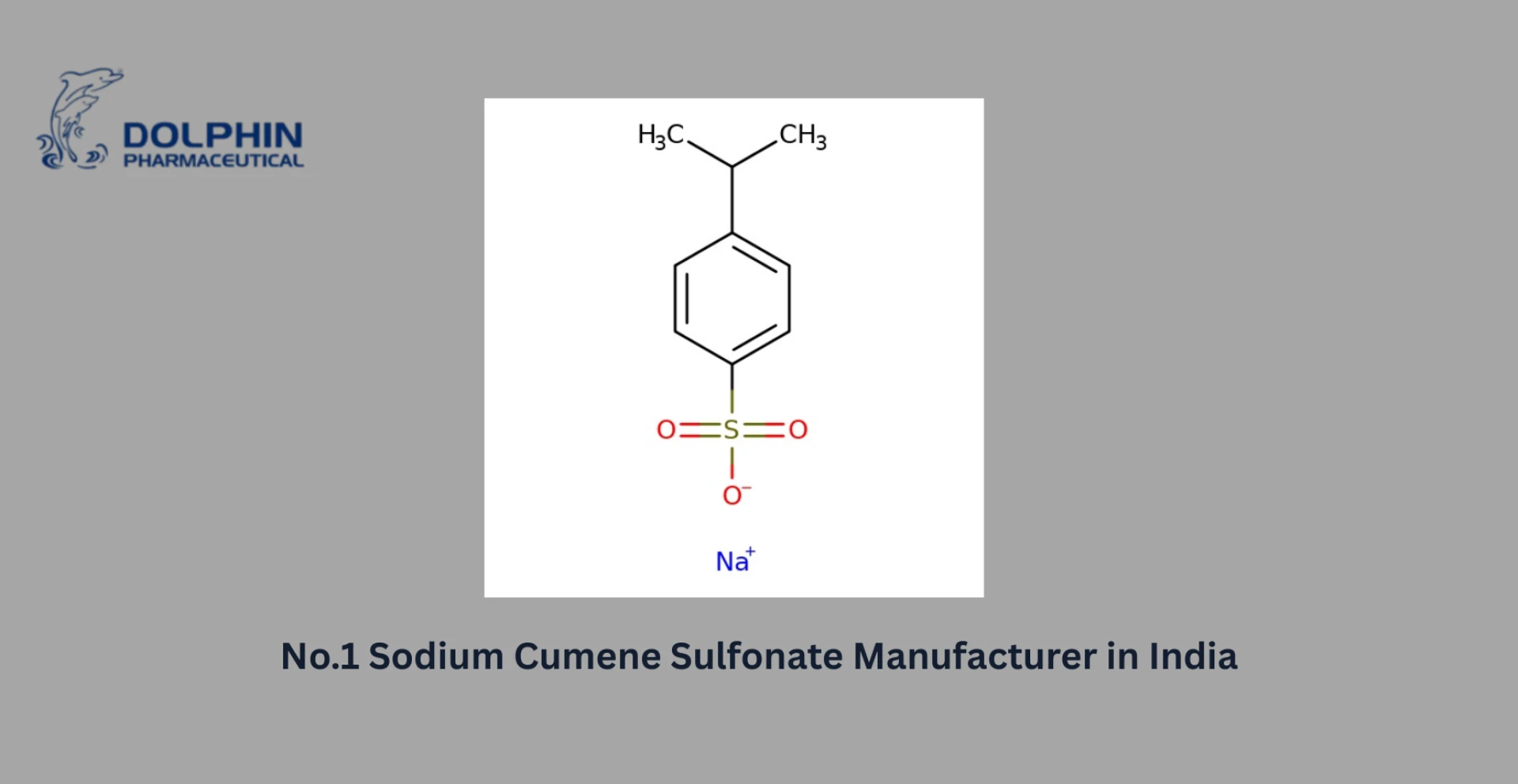
Examples of Commonly Used Pharma Intermediates
Some widely used pharmaceutical intermediates include:
- 4-Chlorobenzhydryl chloride (used in antihistamines)
- 2-Chlorobenzyl chloride (used in anti-inflammatory drugs)
- Piperazine derivatives (used in anti-psychotic drugs)
These intermediates are vital to producing a wide range of therapeutic drugs used across global healthcare systems.
3. Types of Pharmaceutical Intermediates
Bulk Drug Intermediates (BDIs)
BDIs are large-scale chemical compounds used in the commercial production of APIs. These are commonly used by pharmaceutical drug manufacturers in India who produce generic drugs for global markets.
Custom and Contract Manufacturing Intermediates
Custom intermediates are tailor-made for specific drug molecules. Dolphin Pharmaceutical offers contract manufacturing services for companies seeking specialized solutions, ensuring compliance with their unique chemical and regulatory requirements.
Specialty Intermediates for High-Potency Drugs
High-potency intermediates are used in the manufacture of drugs like oncology therapies and hormonal treatments. These require strict containment protocols and highly skilled teams, areas where Dolphin excels through state-of-the-art infrastructure.
4. Regulatory and Quality Standards for Pharma Intermediates
International Guidelines (ICH, USFDA, EU GMP)
Although pharmaceutical intermediates may not be as heavily regulated as APIs, most intermediates manufacturers in India voluntarily adhere to global standards. Guidelines from ICH, USFDA, and EU GMP ensure the consistency, traceability, and safety of these compounds.
Importance of Traceability and Purity
Traceability helps in tracking the origin and process history of every intermediate. This is essential for recalls, audits, and regulatory filings. Maintaining a high level of purity also minimizes the risk of cross-contamination in the final drug formulation.
Compliance Requirements for Global Supply
To supply pharmaceutical intermediates globally, manufacturers must comply with the importing country’s regulations, submit detailed technical documentation, and undergo regular audits. Dolphin Pharmaceutical ensures end-to-end compliance, making us a trusted name worldwide.
5. How Dolphin Pharmaceutical Ensures High-Quality Intermediates
Advanced R&D and Process Optimization
Our dedicated R&D team continuously works on improving synthetic pathways, reducing waste, and increasing yield. Through innovative research, we develop cost-effective and eco-friendly intermediates suitable for large-scale manufacturing.
In-House Manufacturing and Global Certifications
As both a pharmaceutical drug manufacturer in India and an intermediates manufacturer in India, Dolphin Pharmaceutical operates certified, GMP-compliant facilities. This vertical integration allows for complete quality control and faster turnaround times.
Focus on Sustainability and Safety Standards
We integrate green chemistry practices, reduce solvent usage, and recycle materials wherever possible. Our facilities follow strict safety protocols to protect both our employees and the environment.
6. Global Demand and Market Trends for Pharma Intermediates
Rising Need for Cost-Effective Drug Manufacturing
The global pharmaceutical industry is under pressure to reduce costs without compromising quality. Indian intermediates offer a competitive advantage due to lower manufacturing costs and high production standards.
Growth in Custom Manufacturing and CDMO Services
More global pharma companies are outsourcing intermediate production to intermediates manufacturers in India through CDMO (Contract Development and Manufacturing Organization) partnerships. This trend is expected to grow rapidly in the coming years.
India’s Role as a Global Hub for Intermediates
India is emerging as a global hub for pharmaceutical intermediates, thanks to its strong chemical industry base, skilled workforce, and cost-effectiveness. Dolphin Pharmaceutical proudly contributes to this growth by serving clients across Europe, the Americas, and Asia.
7. Choosing the Right Manufacturer for Pharma Intermediates
Key Criteria: Quality, Reliability, Scalability
Choosing the right partner involves evaluating their manufacturing capacity, certifications, and commitment to quality. A reliable supplier must be transparent, consistent, and capable of scaling up production without compromising standards.
The Advantage of Working with Dolphin Pharmaceutical
With decades of expertise, state-of-the-art facilities, and a global client base, Dolphin Pharmaceutical stands out as a dependable intermediates manufacturer in India. We provide end-to-end solutions with a strong focus on quality, safety, and on-time delivery.
Conclusion
Pharmaceutical intermediates are essential to the safe and efficient production of active pharmaceutical ingredients and final drug formulations. As the industry grows and global standards become more stringent, the need for reliable and high-quality intermediates becomes more critical than ever.
At Dolphin Pharmaceutical, we are proud to lead as a trusted pharmaceutical drug manufacturer in India and intermediates manufacturer in India, delivering innovative, sustainable, and globally compliant solutions that meet the highest quality benchmarks. Our mission is to support global health with excellence, one molecule at a time.

















Sign in to leave a comment.