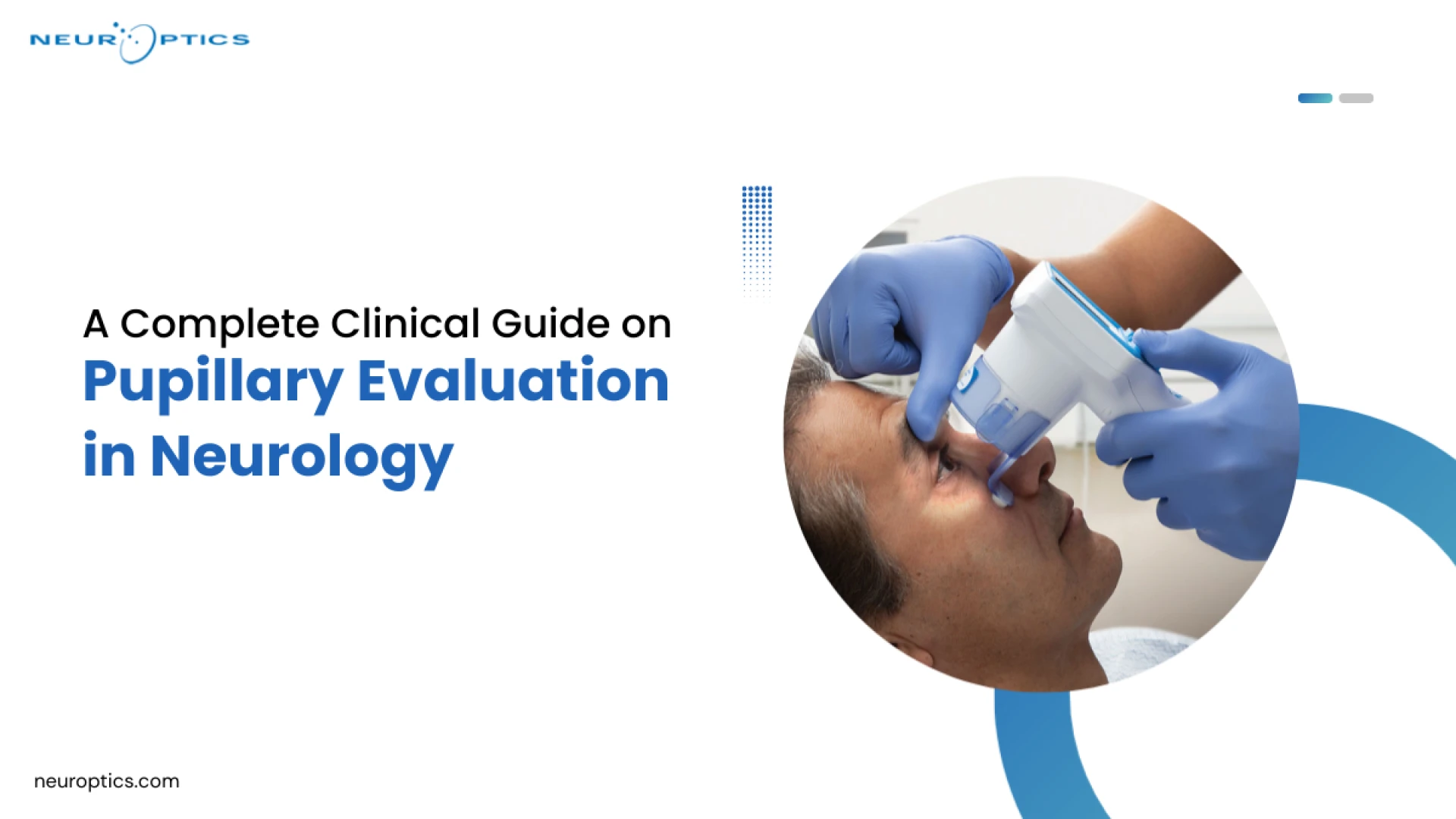
Walk into any neuro-ICU on a weekday morning and watch what happens during rounds. Attendings, residents, and nurses each assess the same patient's pupils — and document three different findings. Not because anyone is wrong, exactly. But because manual neuro exam technique varies by training, experience, and even the quality of the penlight in someone's coat pocket.
That inconsistency has real consequences. Missed trends, delayed escalation, and documentation gaps that don't hold up in adverse event review. And it's one reason why standardized pupillary evaluation has become a growing priority in hospitals that take neurological monitoring seriously.
This guide is written for clinical teams — nurses, residents, intensivists, and neuro-critical care staff, who want a clearer picture of what thorough pupil assessment looks like, why it matters, and where better tools are changing outcomes.
Clinical Significance of Pupillary Findings
Pupils rarely hide what the brain is going through. Their size, symmetry, and responsiveness often mirror what is happening in the underlying neuroanatomy. When a clinician interprets those signals carefully, they gain valuable diagnostic insight—something no triage score by itself can fully reveal.
Anisocoria — a size difference greater than 1 mm between pupils — is benign in a large portion of the general population. In a post-operative neurosurgical patient or someone with a declining GCS, it signals a potential emergency. Context is everything, which is why documentation has to be precise. "Unequal" is not a clinical finding. "Left pupil 5 mm, right pupil 3 mm, left sluggishly reactive" is.
Reactivity tells a parallel story. The pupillary light reflex travels through CN II and CN III, synapsing in the pretectal nucleus of the midbrain. A unilateral, sluggish, or absent response in the right clinical setting points toward CN III compression — the kind associated with uncal herniation or a posterior communicating artery aneurysm. Catching that finding early, before herniation becomes irreversible, is the entire point.
Shape deserves attention too. Oval, teardrop-shaped, or fixed mid-dilated pupils often reflect direct ocular trauma, prior surgery, or acute angle-closure glaucoma. Those findings belong in the chart with specificity — not vague descriptors that mean something different to every provider who reads them.
Limitations of Manual Pupillary Assessment
Honest clinicians will admit that bedside pupil measurement is imprecise. The standard penlight method depends on ambient lighting, examiner technique, and a subjective read of what "brisk" looks like at 3 AM on hour eleven of a shift.
Across multiple providers over a 12-hour period, that variability compounds. A pupil that measured 3.2 mm at 6 AM and 4.1 mm at noon may look entirely normal to each individual examiner in isolation, but that change is clinically meaningful. It's the kind of trend an automated device would flag immediately, and that manual assessment quietly misses.
The downstream effects are real. Delayed recognition of secondary neurological injury. Inconsistent escalation thresholds across care teams. Documentation that captures a single moment rather than a trajectory. None of that serves the patient, and none of it holds up well when outcomes come under review.
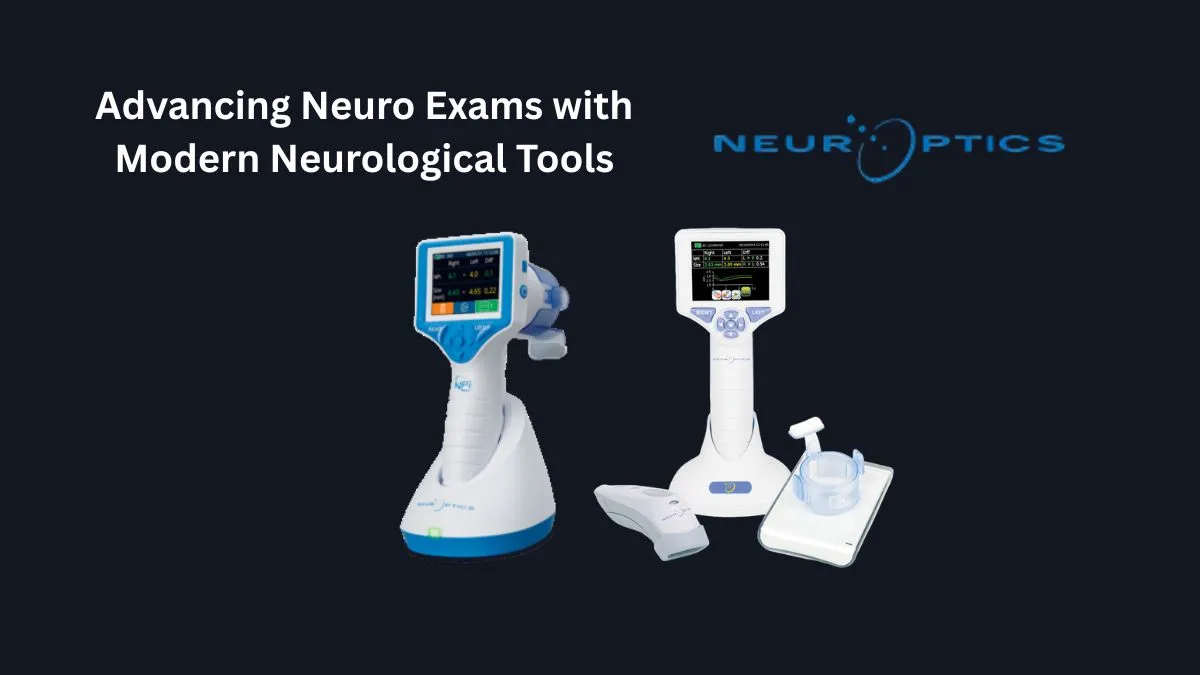
The Role of Automated Pupillometry and NPi

This is where purpose-built neurological tools — specifically infrared automated pupillometers — have shifted the standard of care in high-acuity settings. These devices measure constriction velocity, latency, maximum and minimum diameter, and dilation speed, then generate a composite score called the NPi, on a scale from 0 to 5.
An NPi below 3 is considered abnormal. More importantly, a declining trend — even within the technically normal range — frequently signals the earliest detectable rise in intracranial pressure or evolving secondary injury. That distinction matters enormously. It shifts clinical focus from a single data point to a trajectory, which is where real monitoring value lives.
Consider this scenario: a patient in the neuro-ICU following subarachnoid hemorrhage has stable manual pupil exams documented across 18 consecutive hours. Automated monitoring identifies a gradual automated pupil score decline over a four-hour window. A repeat CT angiogram confirms early vasospasm. Intervention follows before any clinical deterioration is visible. Those four hours represent the entire margin between a good outcome and a permanent deficit.
Standardizing Pupillary Assessment in Clinical Practice
Hospitals that improve pupillary monitoring usually change more than just the equipment. The real improvement often comes from clearer documentation and consistent exam technique.
Start with shared terminology. Words such as PERRL, brisk, sluggish, and absent should be used consistently so that every clinician reading the chart understands the finding the same way.
Training also matters. When teams understand the anatomy behind the pupillary reflex and how pupillary score trends are interpreted, documentation becomes more precise and escalation decisions happen sooner.
Automated monitoring is most useful in higher-acuity settings such as neuro-ICUs, post-cardiac arrest care units, neurosurgical step-down wards, and TBI management units. Using the technology where neurological monitoring is critical keeps the workflow practical while improving consistency across shifts.
Benefits of Objective Pupillary Monitoring in Neurological Care
Reducing secondary neurological injury often leads to shorter ICU stays and lowers the overall cost of treatment per patient. In studies involving people with traumatic brain injury (TBI), researchers have observed that when clinicians intervene earlier using automated pupil score-guided assessments, patients tend to show better functional recovery by the time they are discharged.
Objective, timestamped documentation reduces liability exposure in ways that narrative notes cannot. "pupillary score declined from 3.8 to 2.4 over six hours; neurology was notified at hour three." That kind of precise documentation is far more reliable than a note that simply reads, "pupils sluggish, MD notified. Staff efficiency also improves when clinicians aren't second-guessing ambiguous manual findings or pulling a colleague in for a second look on a borderline exam.
Frequently Asked Questions
Q: Can pupillary findings replace neuroimaging?
A: No. Their value is rapid bedside surveillance and early trend detection; not a substitute for CT or MRI, which answer fundamentally different clinical questions.
Q: What does an NPi score of 0 mean?
A: An absent pupillary light reflex — the most severe finding on the scale. It requires immediate evaluation, ICP correlation, and urgent imaging consideration. It does not independently confirm brain death, which demands a formal protocol.
A: Do medications interfere with findings?
A: Significantly. Opioids cause miosis; anticholinergics produce fixed dilation; sympathomimetics drive mydriasis. Every finding must be interpreted alongside the current medication profile — one more reason objective pupillary score trending outperforms isolated manual snapshots.
Advancing Standards in Neurological Monitoring
Pupillary assessment has always been foundational to neurological care. What's changed is the expectation — for consistency, precision, and the ability to detect deterioration before it crosses an irreversible threshold. Hospitals that standardize their approach and deploy the right tools where acuity demands it see the returns: better outcomes, stronger documentation, and clinical teams that trust what they're seeing.
The starting point is an honest audit of current practice. Where does variability live? What does documentation actually look like across shifts? Answer those questions, and the path forward becomes clear.
Ready to evaluate how automated pupillary monitoring fits your hospital's neurological protocols? Connect with NeurOptics, a clinical specialist, to identify where standardization closes the gaps your current workflow leaves open.















Sign in to leave a comment.