A comprehensive physicochemical property evaluation is an important premise for the development of formulation and process. It provides an important basis for the druggability evaluation of the compound, route of administration and dosage form selection, formulation and process design, and the quality control of the drug product. It is a solid foundation for the development of safe, effective, and quality-controllable medicines.
Relying on the top-notch instruments and technical capabilities, Crystal Pharmatech can provide customers with a comprehensive physicochemical property evaluation.
Excipient Selection and Compatibility Studies
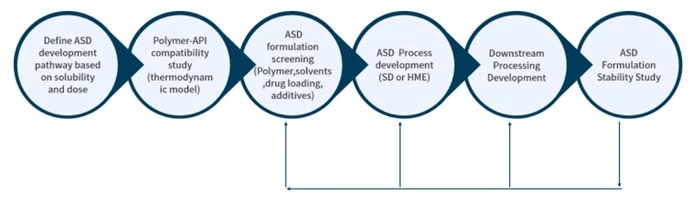
Excipients play an important role in drug release, drug stability and manufacture of drug product. We help customers select the appropriate excipients in early formulation development through screening and technical know-how to investigate the interactions between API and excipients. The excipient selection will be tailored to the individual API depending on physicochemical properties of API, indication, route of administration, species and so on. By optimizing pre formulation, bioavailability and exposure levels can be improved.
Formulations for PK/Efficacy/pre formulation study
Contrary to popular belief, early pre formulation development is not simply dumping API in a vehicle and gavage feeding an animal. If you do this, you will most likely lose time and money with unnecessary and confusing PK studies. This led to the development of our SMART biopharmaceutical solutions that offer input from our scientific advisory board: A one-stop shop from candidate selection through GMP FIH.
As a professional cdmo pharma company, crystal biotech trys our best to provide good products and service for you.
For more information about contract research pharma, please feel free to contact us!
















Sign in to leave a comment.