(Synonyms: MC-VC-PAB-MMAE, Vedotin)
Catalog No.GC37891
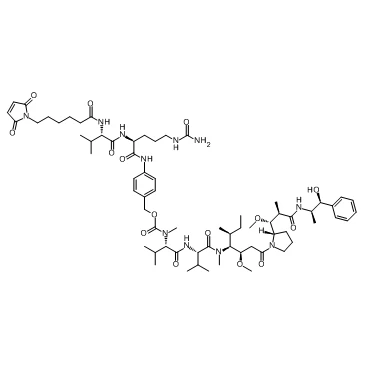
VcMMAE is a drug-linker conjugate used in the synthesis of antibody-drug conjugates (ADCs), consisting of the microtubule inhibitor monomethyl auristatin E (MMAE) and a cleavable valine-citrulline (vc) linker.
Products are for research use only. Not for human use. We do not sell to patients.
Description
VcMMAE is a drug-linker conjugate used in the synthesis of antibody-drug conjugates (ADCs), consisting of the microtubule inhibitor monomethyl auristatin E (MMAE) and a cleavable valine-citrulline (vc) linker[1-2]. In the ADC structure, VcMMAE is covalently linked to the antibody via the maleimide group of its linker. Upon specific cleavage by cancer cell lysosomes, VcMMAE releases the cytotoxic MMAE, thereby inhibiting tubulin polymerization and inducing tumor cell apoptosis[3-4].
In vitro, when conjugated with the anti-HER2 antibody H32 (0.02–0.78nM) and applied to HER2-positive cancer cells such as N87, SK-BR-3, and BT474 for 16 hours, VcMMAE significantly inhibited cancer cell viability and induced apoptosis[5]. The conjugate of VcMMAE with Rituximab (100–1000ng/mL) treatment of CD20-positive Raji cells for 48–72 hours markedly reduced cell viability and induced cell death[6].
In vivo, the conjugate of VcMMAE with the chimeric monoclonal antibody cAC10 targeting CD30 (1-30mg/kg), administered via tail vein injection every 4 days to tumor-bearing SCID mice, significantly suppressed the growth of CD30-positive anaplastic large cell lymphoma (Karpas 299) and Hodgkin's disease (L540cy) xenografts, leading to complete tumor regression[7]. The conjugate of VcMMAE with the anti-CD22 monoclonal antibody HB22.7 (7.5mg/kg), administered as a single intraperitoneal injection to tumor-bearing ICR-SCID mice, significantly inhibited the growth of DoHH2 (transformed follicular lymphoma) and Granta 519 (mantle cell lymphoma) xenografts and induced complete tumor regression[8].
References:
[1] Best RL, LaPointe NE, Azarenko O, et al. Microtubule and tubulin binding and regulation of microtubule dynamics by the antibody drug conjugate (ADC) payload, monomethyl auristatin E (MMAE): Mechanistic insights into MMAE ADC peripheral neuropathy. Toxicol Appl Pharmacol. 2021 Jun 15;421:115534.
[2] Pollack VA, Alvarez E, Tse KF, et al. Treatment parameters modulating regression of human melanoma xenografts by an antibody-drug conjugate (CR011-vcMMAE) targeting GPNMB. Cancer Chemother Pharmacol. 2007 Aug;60(3):423-35.
[3] Vaklavas C, Forero A. Management of metastatic breast cancer with second-generation antibody-drug conjugates: focus on glembatumumab vedotin (CDX-011, CR011-vcMMAE). BioDrugs. 2014 Jun;28(3):253-63.
[4] Zhang J, Li M, Li W, et al. Mechanistic Insights into Anti-Nectin4-VcMMAE-Induced Ocular Toxicity: From Cellular Uptake Pathways to Molecular Modification. Int J Mol Sci. 2025 May 22;26(11):4996.
[5] Chiang ZC, Chiu YK, Lee CC, et al. Preparation and characterization of antibody-drug conjugates acting on HER2-positive cancer cells. PLoS One. 2020 Sep 28;15(9):e0239813.
[6] Abdollahpour-Alitappeh M, Hashemi Karouei SM, Lotfinia M, et al. Amanzadeh A, Habibi-Anbouhi M. A developed antibody-drug conjugate rituximab-vcMMAE shows a potent cytotoxic activity against CD20-positive cell line. Artif Cells Nanomed Biotechnol. 2018;46(sup2):1-8.
[7] Francisco JA, Cerveny CG, Meyer DL, et al. cAC10-vcMMAE, an anti-CD30-monomethyl auristatin E conjugate with potent and selective antitumor activity. Blood. 2003 Aug 15;102(4):1458-65.
[8] Abuhay M, Kato J, Tuscano E, et al. The HB22.7-vcMMAE antibody-drug conjugate has efficacy against non-Hodgkin lymphoma mouse xenografts with minimal systemic toxicity. Cancer Immunol Immunother. 2016 Oct;65(10):1169-75.
Synonyms: MC-VC-PAB-MMAE, Vedotin
vcmmae
More from glpbio
View all →More in Biotech
Browse all in Biotech → Biotech
Biotech
Why Do Western Blotting Steps Fail Despite Proper Setup?
 Biotech
Biotech
Diving Deep: Exploring the World of Underwater Construction Services
 Biotech
Biotech
JFK Airport Departures Guide – Live Flights, Terminals, Delays & Travel …
 Biotech
Biotech
How to Choose the Best LIMS Software in India (Complete Guide)
 Biotech
Biotech
The Magic of Water Soluble Polymers: Sustainable Solutions for a Greener…
 Biotech
Biotech
Industrial Quartz Glass for Precision Applications
Sign in to leave a comment.