Interferon signaling is one of the most critical defense mechanisms cells use against viral infection and immune disruption. At the center of this pathway is STAT1 (Signal Transducer and Activator of Transcription 1), a transcription factor that converts extracellular cytokine signals into gene-level responses.
If you’re studying immune signaling without directly measuring STAT1 activity, your analysis is incomplete. STAT1 antibodies exist to solve exactly that problem—allowing precise detection, localization, and functional analysis of this protein across experimental systems.
Why STAT1 Detection Actually Matters
STAT1 is not just present or absent—it shifts between inactive and active states depending on phosphorylation. Once activated, it translocates to the nucleus and initiates transcription of interferon-stimulated genes.
This creates a common mistake in research:
- Measuring total STAT1 and assuming pathway activation
- Ignoring phosphorylation status (which is what actually matters)
A properly validated STAT1 antibody lets you distinguish between these states, which is the difference between real signaling data and misleading conclusions.
Molecular Basis: What You’re Really Measuring
STAT1 antibody performance depends on one thing: epitope recognition.
- Antibodies bind to specific amino acid sequences or structural regions
- High specificity ensures STAT1 is detected—not related STAT proteins
- Phospho-specific antibodies target activated STAT1 forms (e.g., Tyr701)
If epitope targeting is poorly characterized, cross-reactivity becomes a real issue—and your results become unreliable.
Production also matters more than people admit:
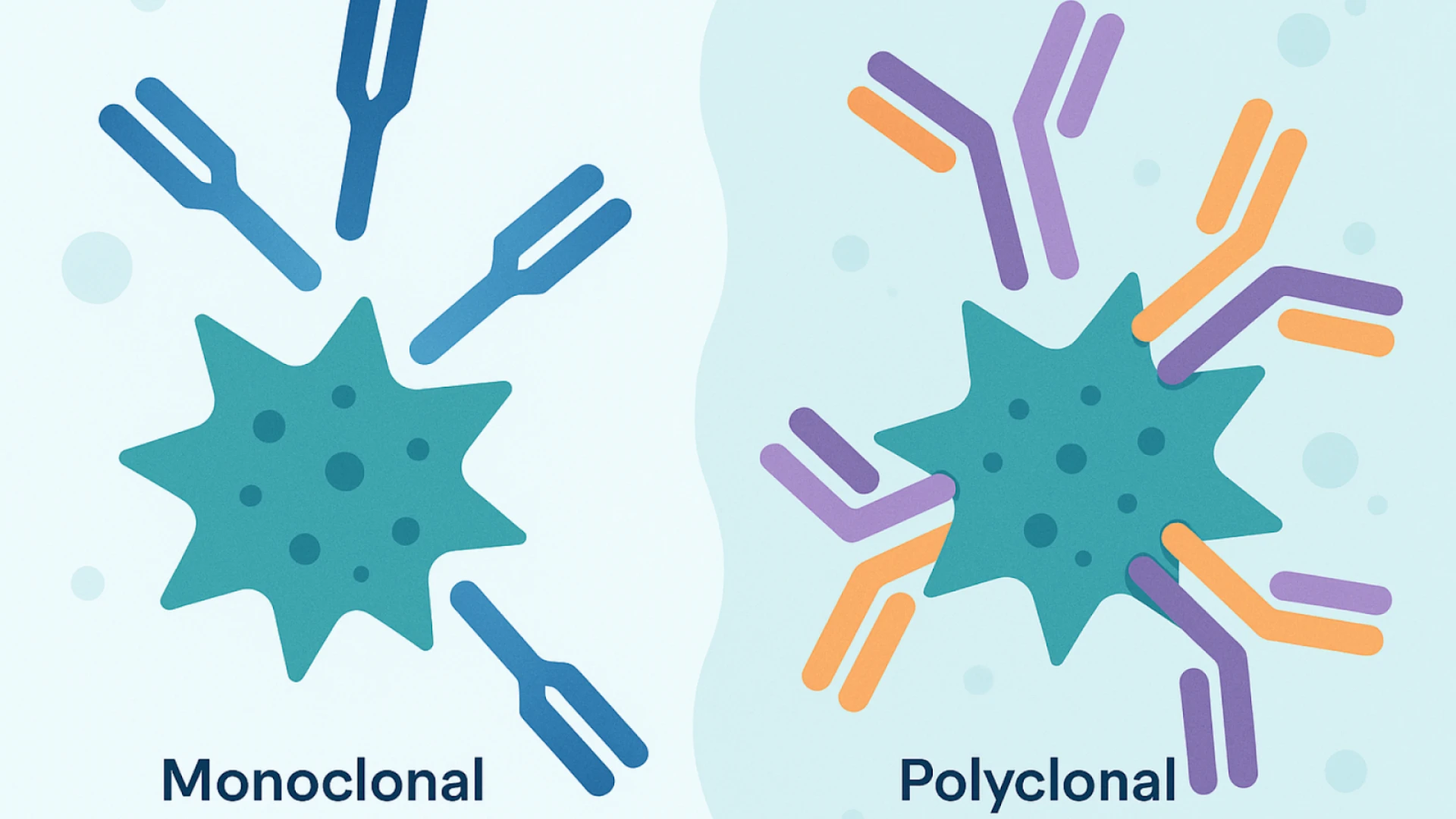
- Monoclonal antibodies → consistent, specific, reproducible
- Polyclonal antibodies → more sensitive but less consistent
If your experiments require repeatability, monoclonals are usually the safer choice.
Where STAT1 Antibodies Are Actually Used
Western Blotting
Still the baseline technique, but often misused.
With STAT1 antibodies, Western blotting allows you to:
- Detect total STAT1 levels
- Measure phosphorylation (activation state)
- Confirm expected protein size
If your band pattern is inconsistent, it’s usually bad technique or poor antibody validation—not biology.
Immunofluorescence Microscopy
This answers a question Western blot cannot: where is STAT1?
- Cytoplasmic STAT1 → inactive
- Nuclear STAT1 → active
Immunofluorescence enables:
- Visualization of nuclear translocation
- Co-localization with other signaling proteins
- Spatial understanding of pathway activation
Ignoring localization leads to shallow conclusions.
Flow Cytometry
This moves from single-sample analysis to population-level insight.
STAT1 antibodies in flow cytometry help:
- Quantify signaling across thousands of cells
- Detect variability within immune populations
- Measure response to cytokine stimulation
If you care about heterogeneity (and you should), this is essential.
Immunoprecipitation and Protein Interaction Studies
STAT1 operates within complexes, not in isolation.
Using immunoprecipitation:
- STAT1 can be pulled down with interacting proteins
- Binding partners can be identified
- Signaling networks can be mapped
If you’re only measuring expression but not interactions, you’re oversimplifying the biology.
ELISA for Quantitative Analysis
For studies that require quantification rather than visualization:
- ELISA enables measurement of STAT1 levels across samples
- High-throughput format supports large datasets
- Useful in biomarker and drug-response studies
However, ELISA is only as reliable as the antibody used—poor specificity ruins quantification.
Experimental Reality: Where Most Data Falls Apart
Let’s cut the polite version—most signaling data is flawed because of basic mistakes:
- Wrong antibody concentration → weak signal or high background
- No proper controls → results are not interpretable
- No validation → assuming specificity without proof
- Inconsistent sample prep → degraded or altered protein
Minimum standards you should follow:
- Use positive and negative controls
- Validate with knockdown or knockout systems
- Include loading controls for normalization
- Replicate across conditions
If you skip these, your conclusions don’t hold up.
Validation Is Not Optional
A “working antibody” is not the same as a validated one.
Proper validation includes:
- Testing across multiple applications (WB, IF, flow)
- Confirming specificity via genetic knockdown
- Comparing different antibody clones
- Cross-validating with independent techniques
If this isn’t documented, you’re taking a gamble.
Bottom Line
STAT1 antibodies are not just routine lab reagents—they’re critical tools for decoding interferon signaling and immune response mechanisms. But their value depends entirely on how rigorously they’re validated and how carefully they’re used.
If you:
- Don’t distinguish total vs phosphorylated STAT1
- Don’t validate specificity
- Don’t control your experiments properly
Then your data isn’t just weak—it’s potentially wrong.
Reliable signaling research comes down to disciplined methodology, not just good intentions or expensive reagents.

















Sign in to leave a comment.