A rigorous look at epigenetic clocks, mTOR biology, and whether an Easter Island soil fungus holds the key to rewinding time.
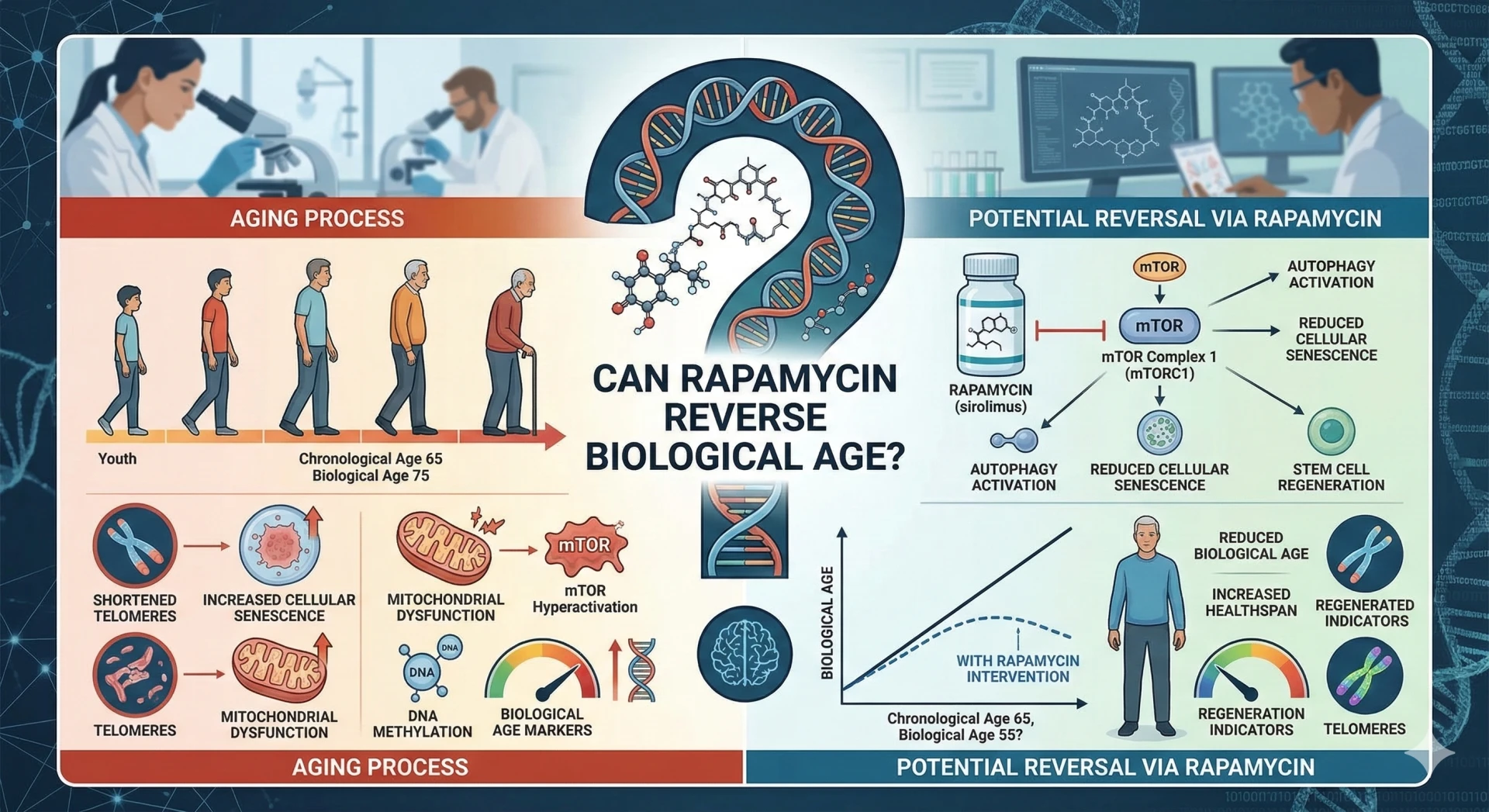
In 2009, a landmark finding changed longevity science: rapamycin — derived from a bacterium in Easter Island soil — extended median lifespan in mice by up to 14%, even when treatment began late in life. It was the first drug to robustly extend mammalian lifespan. But the more radical question has since emerged: can rapamycin do something even more striking and actually reverse the biological clock?
To answer that, researchers have turned to epigenetic clocks — methylation-based biomarkers that, arguably better than chronological age, capture how worn and weathered the body truly is. The question is no longer theoretical. With self-experimenters dosing weekly, randomized trials recruiting, and dog studies accumulating data, the evidence landscape is expanding fast — even if clarity has not yet followed.
This piece examines the mechanistic case, the actual data from cells to humans, what leading experts say, what real users experience, and what an honest risk-benefit picture looks like for anyone seriously exploring this space.
What Are Epigenetic Clocks, and Why Do They Matter?
Your chronological age is just a number. Your biological age — how fast your cells are actually deteriorating — is what determines your risk of disease, cognitive decline, and death. Epigenetic clocks are the most sophisticated tools we currently have for measuring that biological age.
These clocks work by tracking DNA methylation: chemical tags (methyl groups) that accumulate at specific sites (CpG positions) across the genome in predictable patterns as we age. The Horvath pan-tissue clock, published in 2013, was the first to demonstrate that a mathematical model built from these methylation patterns could predict biological age across virtually every tissue in the body — and that biological age predicted mortality risk better than chronological age alone.
Second-generation clocks like GrimAge and DunedinPACE (the "pace of aging" clock) go further, measuring not just current biological age but how fast someone is aging in real time — a distinction with profound implications for evaluating interventions.
Does Rapamycin Slow Epigenetic Aging? What the Studies Show
The 2019 Skin Cell Study: A Clear Preclinical Signal
The foundational human-cell study came in 2019, when Horvath's group treated cultured human skin fibroblasts with rapamycin and measured the pan-tissue epigenetic clock. The drug significantly slowed the rate at which methylation age was advancing — and did so independently of cell proliferation rate or senescence status. This was important: it suggested rapamycin wasn't simply slowing the clock by slowing cells down, but by directly altering the epigenetic machinery.
Genome-wide association data bolstered this finding: human variants in the mTOR signaling pathway are significantly associated with faster pan-tissue epigenetic clock advancement in population-level analyses, suggesting the mTOR-methylation relationship is a biologically embedded feature of human aging, not a lab artifact.

Mouse Data: Strong Lifespan Results, Mixed Clock Data
In mice, rapamycin's lifespan effects are the strongest ever recorded for a pharmacological agent: 9% in males and up to 14% in females in the NIA Interventions Testing Program, beginning treatment at the human equivalent of ~60 years old. Subsequent mouse research has shown that rapamycin prevents 20–40% of age-related methylation changes in the brain — a striking result, though tissue-specific clock data from other organs remains more mixed.
A series of studies from 2020–2023 demonstrated that short-course rapamycin treatment (10 weeks) rejuvenated periodontal bone density, cardiac function, and gut microbiome composition in aged mice, lending biological plausibility to epigenetic benefits even from intermittent treatment windows.
Human Studies: The Honest Picture
Here is where the evidence gets thin. No large-scale randomized controlled trial has yet confirmed that rapamycin reverses systemic epigenetic age in healthy humans. The human data we do have comes from two sources:
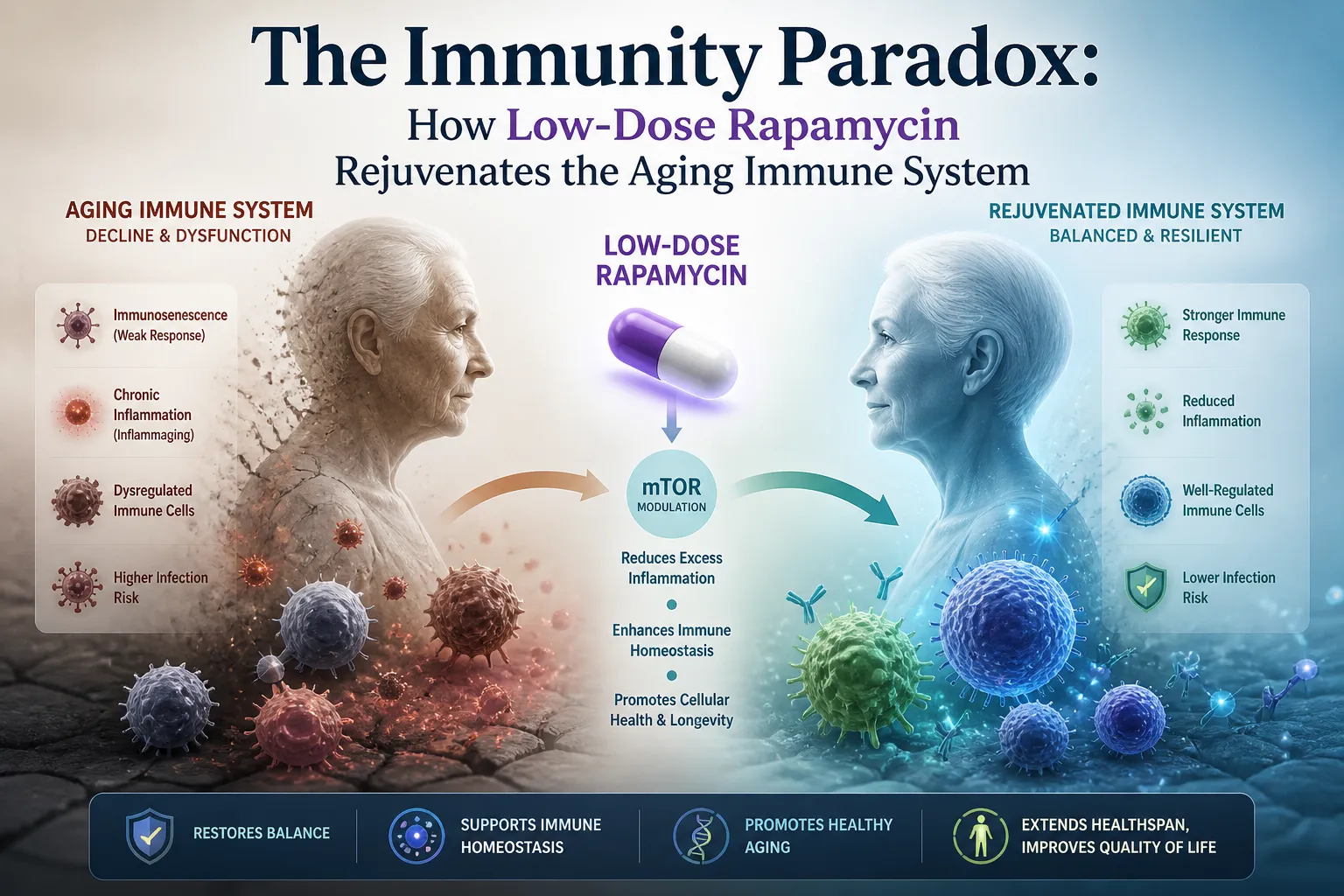
Immune function studies: Joan Mannick's trials of the rapalog RAD001 (everolimus) in elderly patients showed ~20% improvement in influenza vaccine response — compelling evidence of immune rejuvenation, but without epigenetic clock endpoints.
Topical rapamycin: A small trial applying rapamycin cream to skin measured local epigenetic clock reversal over six months, with promising but limited results.
The key trials now underway include the PEARL trial (AgelessRx) — a randomized placebo-controlled study in healthy adults with epigenetic clocks as primary endpoints — and the Dog Aging Project's TRIAD trial, widely considered the pivotal translational bridge between rodent data and human medicine.
The Mechanism: How mTOR Inhibition Could Reset the Epigenome
Rapamycin works by inhibiting mTORC1 — the mechanistic target of rapamycin complex 1 — a master metabolic sensor that integrates nutrient signals, growth factors, and cellular stress into decisions about whether to grow, reproduce, or maintain and repair. When mTORC1 is overactive (a hallmark of aging and metabolic disease), it accelerates a cascade of damage. When blunted, the cell shifts toward longevity-associated programs.
The epigenetic connection runs through several overlapping pathways:
Autophagy upregulation: mTOR inhibition triggers autophagy — the cellular "self-cleaning" system that removes damaged proteins and organelles. Reducing this inflammatory burden appears to slow methylation drift at clock CpG sites.
DNMT and TET enzyme modulation: Rapamycin alters the activity of DNA methyltransferases (writers of methylation marks) and TET enzymes (erasers), the direct molecular machinery that epigenetic clocks measure.
Reduction of stochastic epigenetic noise: mTOR inhibition reduces the rate of random histone exchange and transcription-driven methylation errors that accumulate over time — a process some researchers call "epigenetic entropy."
Mitochondrial and stem cell effects: Matt Kaeberlein notes that rapamycin's epigenetic benefits appear distinct from telomere attrition pathways, likely involving mitochondrial quality control and stem cell preservation.
In keratinocyte experiments, rapamycin's effects on epigenetic age were decoupled from its effects on cellular senescence — suggesting broad applicability across cell types and potentially across tissues in a whole organism.
What Leading Experts Actually Say
Matt Kaeberlein — The Translational Realist
Kaeberlein is one of rapamycin's most rigorous scientific advocates, and one of its most careful communicators. He consistently pushes back on overclaiming, emphasizing that epigenetic clocks are one biomarker among many, and that the TRIAD trial represents the necessary next step before drawing conclusions about human aging. His view: intermittent dosing likely preserves benefits while minimizing immune risk — but this needs to be tested, not assumed.
Peter Attia — The Clinical Optimist
Attia has been vocal about his own low-dose intermittent rapamycin use and covers it extensively in his writing and podcast. He highlights the extraordinary late-life efficacy in mice (some studies showing up to 60% lifespan extension in late-starting protocols), and calls for optimized human protocols around timing, frequency, and dose. His position: the animal data is compelling enough to warrant careful personal experimentation under medical supervision, while awaiting trial results.
Joan Mannick — The Clinical Evidence
Mannick's clinical work with RAD001 (everolimus) in elderly patients provided the clearest human evidence that selective mTORC1 inhibition can restore immune function without severe immunosuppression. Her consistent emphasis: the dosing regimen matters enormously. Continuous dosing mimics transplant protocols with their attendant risks; intermittent or cyclical dosing may access longevity benefits through a fundamentally different mechanism.
Scientific Consensus — Honestly Stated
The honest scientific consensus in 2025 is this: rapamycin is the most mechanistically credible pharmacological candidate in longevity science, with the strongest animal data of any known intervention. It is not a proven intervention for reversing human biological age. The gap between those two statements is where the legitimate research effort is currently directed.
Real-World User Experiences: What Self-Experimenters Report
The longevity community has generated a substantial body of n-of-1 data on rapamycin — mostly anecdotal, often confounded, but still informative about the real-world experience of intermittent dosing.
Variable epigenetic clock results: Some self-experimenters report 1–4 year reductions in biological age on commercial tests like TruAge or Levine's PhenoAge after 6–12 months of use. Others see no change. Confounders — simultaneous lifestyle optimization, placebo response — make individual results nearly uninterpretable.
Functional benefits: More consistent reports include improved workout recovery time, subjective energy, and reduced inflammatory symptoms. These align loosely with the immune and mitochondrial mechanisms in the literature.
Bryan Johnson's experience: The tech entrepreneur's highly documented biohacking protocol initially included rapamycin, but he discontinued it after observing signals of skin infection susceptibility and what he interpreted as inflammatory clock acceleration in pre-publication biomarker data. His case illustrates both the value of rigorous self-monitoring and the limits of n-of-1 interpretation.
Dosing patterns: Most informed users report 6–10 mg weekly, often with 6–8 week on/4–6 week off cycles. Some add grapefruit juice to increase bioavailability via CYP3A4 inhibition — a practice that effectively multiplies dose unpredictably and is generally not recommended.
Community forums consistently flag the importance of blinded re-testing before drawing conclusions from epigenetic clock changes, and the difficulty of separating rapamycin's effects from the simultaneous lifestyle interventions most serious longevity self-experimenters pursue.
The Risk Side of the Ledger: What You Need to Know
Rapamycin originated as a transplant immunosuppressant — used at daily, high doses to prevent organ rejection. Longevity protocols use doses roughly 10–20x lower, typically intermittently. The risk profile is meaningfully different, but not absent.
Mouth sores (aphthous ulcers): The most commonly reported side effect, appearing in a significant minority of users at 6mg+ weekly. Dose-dependent; often resolves with dose reduction or cycling.
Impaired wound healing: mTORC1 inhibition slows tissue repair. Most protocols recommend ceasing rapamycin several weeks before any planned surgery.
Metabolic effects: Prolonged continuous dosing can induce insulin resistance via mTORC2 inhibition. Intermittent protocols appear to minimize this risk, but long-term data is absent.
Infection susceptibility: Clear immunosuppression at transplant doses. The extent of immune risk at low intermittent doses in healthy adults is genuinely uncertain and probably low, but not zero.
Dyslipidemia: Elevated triglycerides and lipid changes reported in some users and clinical studies. Regular lipid monitoring is standard in informed protocols.
Male fertility: Animal data suggests effects on spermatogenesis. Human implications are poorly characterized. Most practitioners recommend cessation when trying to conceive.
Unknown long-term effects: There is simply no multi-decade safety data on intermittent low-dose rapamycin in healthy adults. This is not a solvable gap without time.
A Starting Framework for Informed Exploration
If you are considering rapamycin under medical supervision, the following framework reflects current best practices from the scientific literature and informed clinical practitioners. This is not a prescription or recommendation — it is a distillation of what the evidence currently supports.
- Establish a baseline. Before starting, obtain a comprehensive blood panel (lipids, glucose, CBC, inflammatory markers) and an epigenetic clock test using a validated assay (Horvath clock, DunedinPACE, or GrimAge).
- Start low. 3–5 mg weekly is the conventional starting point. Some practitioners favor 6 mg but note higher side effect rates.
- Cycle the dose. 8 weeks on, 4–6 weeks off is a common cycle pattern, based on theoretical reasoning about mTORC2 sparing. No head-to-head RCT has validated this specific pattern.
- Monitor quarterly. Lipid panel, fasting glucose, and CBC at minimum. More comprehensive aging biomarker panels (IL-6, CRP, ferritin) are increasingly used in well-monitored protocols.
- Retest epigenetic clocks at 6 months. This gives enough time for meaningful methylation changes to accumulate, while being early enough to course-correct if adverse biomarker signals appear.
- Evaluate holistically. Clock changes should be evaluated alongside functional markers — not in isolation. A two-year clock reduction alongside rising inflammatory markers may not represent net benefit.
Matt Kaeberlein and other researchers consistently emphasize: the dog aging trials are generating exactly the kind of structured, controlled, endpoint-driven data that the field needs. Following those results before escalating personal protocols is a rational approach.
Conclusion: Where the Science Actually Stands
The mechanistic case is the strongest in longevity pharmacology. The cell culture and mouse data for rapamycin slowing and partially reversing epigenetic clocks are robust. The pathway biology is coherent. The human immune data is encouraging. The expert consensus points in a consistent direction.
What is missing is rigorous, replicated, controlled human data with epigenetic clocks as primary endpoints — and a long enough time horizon to assess whether clock changes actually track with functional health outcomes. That data is coming, slowly, through PEARL, TRIAD, and the broader longevity trial infrastructure that is finally being built.
For now: rapamycin is the most scientifically serious pharmacological candidate for slowing biological aging. It is not a proven intervention for reversing human epigenetic age. Approaching it with curiosity, rigorous monitoring, and genuine epistemic humility about the limits of current evidence is the most defensible posture — for researchers, clinicians, and self-experimenters alike.

Q1: Does rapamycin actually reverse epigenetic age in humans?
Not definitively confirmed. The strongest evidence comes from cell culture (human fibroblasts and keratinocytes) and mouse models, where rapamycin meaningfully slows and partially reverses methylation clock advancement. In humans, small trials — including topical rapamycin on skin — show localized promise, and some self-experimenters report clock reductions on commercial tests. However, no large-scale randomized controlled trial with epigenetic endpoints has yet been completed. The PEARL trial is expected to provide the first rigorous answer.
Q2: How does rapamycin affect the epigenetic clock mechanistically?
Rapamycin inhibits mTORC1, shifting cells away from growth and toward repair and maintenance. This reduces stochastic methylation drift (random epigenetic errors that accumulate with age), upregulates autophagy (reducing inflammatory burden that drives clock acceleration), and modulates the DNMT and TET enzymes that directly write and erase the methylation marks that clocks measure. In mouse brains, this translated to 20–40% fewer age-related methylation changes.
Q3: What is the best epigenetic clock to track rapamycin's effects?
For monitoring purposes, most practitioners recommend a combination approach. DunedinPACE measures the current pace of aging (how fast you are aging right now), making it the most sensitive tool for detecting whether an intervention is working in real time. GrimAge is the strongest predictor of mortality and healthspan. The original Horvath pan-tissue clock has the most historical validation and corresponds most directly to the cell culture data. Retesting after at least 6 months of consistent protocol use gives the most interpretable results.
Q4: What dose of rapamycin do longevity-focused protocols use?
The most commonly discussed range is 3–10 mg weekly, taken intermittently rather than daily. This is substantially lower than transplant protocols (which use daily dosing at much higher levels). A common starting approach is 5 mg weekly, cycling 8 weeks on and 4–6 weeks off. Some practitioners use grapefruit juice to enhance bioavailability via CYP3A4 inhibition, but this makes dosing unpredictable and is generally not recommended for this reason.
Q5: What are the main side effects to watch for?
The most common at longevity doses are mouth sores (aphthous ulcers), which are dose-dependent and usually resolve with dose reduction; impaired wound healing; and elevated triglycerides. Infection susceptibility is a theoretical concern given rapamycin's immunosuppressive mechanism, though at low intermittent doses in healthy adults the risk appears low. Insulin resistance is a risk with continuous high-dose use but appears reduced with intermittent protocols. Regular blood monitoring (lipids, glucose, CBC) is standard practice.
Q6: Did Bryan Johnson stop taking rapamycin, and why?
Yes. Bryan Johnson discontinued rapamycin after his highly documented protocol showed signals he interpreted as concerning — including skin infection susceptibility and what he described as inflammatory biomarker changes. His experience illustrates both the value of rigorous self-monitoring and the difficulty of interpreting n-of-1 data. His case is frequently cited in longevity communities as a reason for caution, though his protocol included many simultaneous interventions making rapamycin-specific conclusions difficult to draw.
Q7: What is the Dog Aging Project TRIAD trial, and why does it matter?
TRIAD (Treatment with Rapamycin for Aging in Dogs) is a randomized controlled trial testing oral rapamycin in over 580 companion dogs across multiple sites. Dogs are considered a uniquely valuable translational model because they are large, long-lived mammals who share human environments, develop many of the same age-related diseases, and have a pace of aging that allows for multi-year studies. The trial measures cardiac function, epigenetic clocks, and a range of aging biomarkers. Experts like Matt Kaeberlein consider TRIAD the most critical near-term test of whether rapamycin's benefits in rodents translate to mammals that resemble humans more closely.
Q8: Can rapamycin be combined with other longevity interventions?
In practice, many longevity-focused physicians combine rapamycin with metformin (for metabolic health), statins (to counteract rapamycin's lipid effects), and comprehensive lifestyle optimization. Some practitioners note theoretical synergies between rapamycin and caloric restriction or intermittent fasting, as both target the mTOR pathway through different mechanisms. However, the combination space is entirely understudied in rigorous trials. Combination protocols add complexity to side effect monitoring and make it harder to attribute effects to any single intervention.
Q9: Is rapamycin safe for long-term use in healthy adults?
The honest answer is that we do not know. Short-term (1–2 year) use at low intermittent doses has an acceptable side effect profile in the self-experimenter and small trial data available. But there is no multi-decade safety data for this use case. The long-term effects on immune function, metabolic health, cancer risk, and wound healing in healthy adults remain genuinely uncertain. This uncertainty is not a reason to dismiss rapamycin, but it is a reason to approach it with appropriate caution, rigorous monitoring, and medical supervision — not as a consumer supplement.
Q10: Where will the most important new data come from?
The PEARL trial (AgelessRx) will be the first placebo-controlled RCT in healthy adults with epigenetic clocks as primary endpoints — expected to report results in 2025–2026. TRIAD results from the Dog Aging Project will provide the critical large-mammal translational test. Second-generation epigenetic clock analyses on existing rapamycin datasets using higher-resolution clocks (including pan-mammalian clocks under development at Altos Labs) may also substantially advance understanding. The broader TAME-like infrastructure for longevity trials, if funded adequately, will eventually enable the definitive human studies the field needs.


















Sign in to leave a comment.