When most people think about living longer, they think about eating well, exercising regularly, and getting enough sleep. These habits matter enormously. But deep inside the laboratories of geroscience, researchers are increasingly focused on something far more specific: a molecule called rapamycin — and the extraordinary evidence that it may be one of the most powerful anti-aging compounds ever discovered.
This is not a fringe idea. Rapamycin is an FDA-approved drug with decades of clinical use. It has extended lifespan in every animal model it has been tested in. And it has produced real, measurable rejuvenation of the immune system in a human clinical trial.
In this article, we'll break down exactly what rapamycin is, how it works, what the science says about its benefits, and why leading researchers believe it may fundamentally change how we approach aging.
What Is Rapamycin? A Brief Background
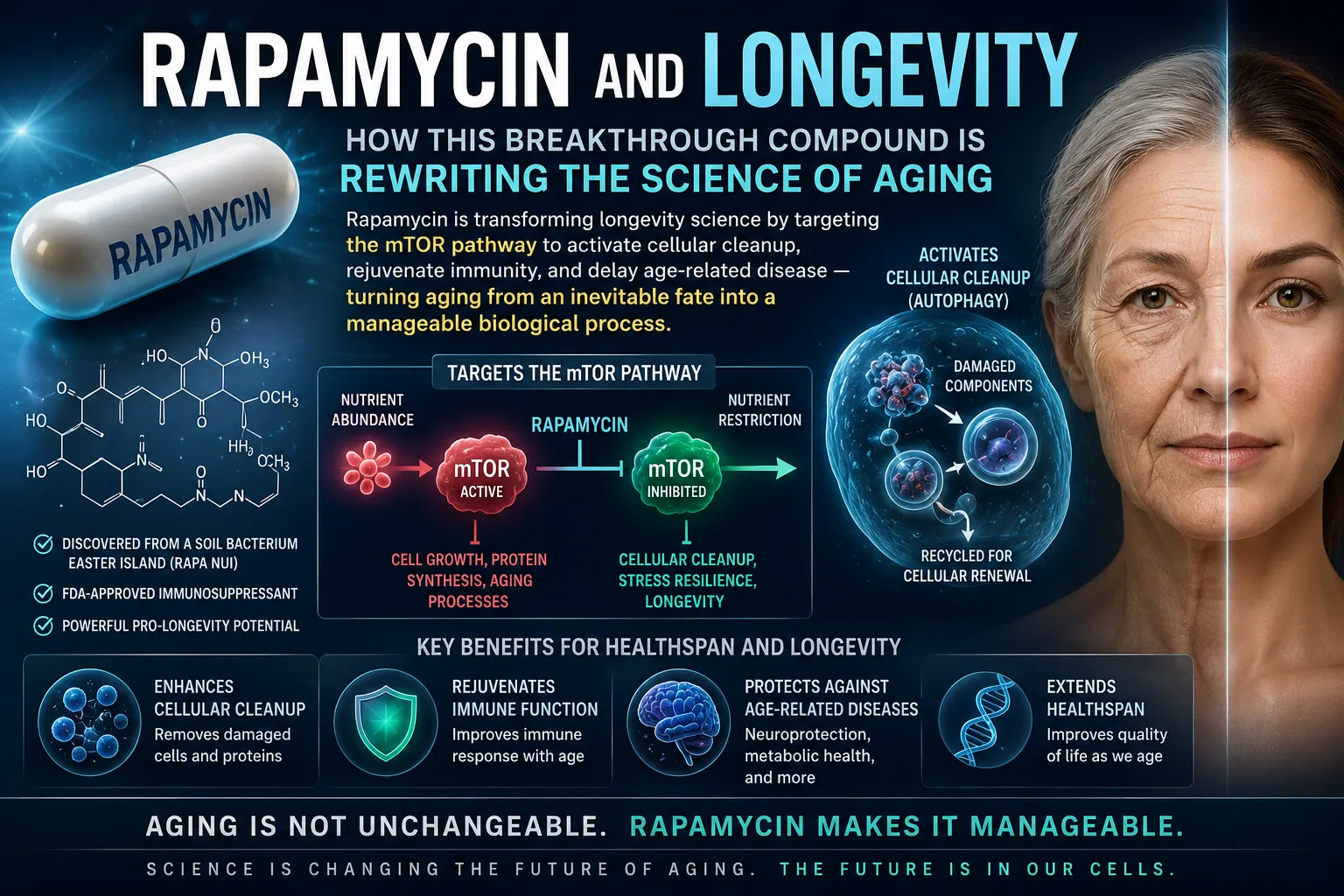
Rapamycin — also known by its generic name sirolimus — is a macrolide compound first isolated in the 1970s from a soil bacterium called Streptomyces hygroscopicus, discovered on Easter Island (Rapa Nui, from which the drug takes its name).
Initially developed as an antifungal agent, rapamycin was later found to have potent immunosuppressive and antiproliferative properties. The U.S. Food and Drug Administration approved it in 1999 to prevent organ rejection in transplant patients — and that clinical application gave researchers decades of safety data.
But it was a landmark 2009 study published in Nature that truly put rapamycin on the map in aging science. Researchers from the National Institutes of Health's Interventions Testing Program demonstrated that feeding rapamycin to aged mice — animals equivalent to 60-year-old humans — extended their median lifespan by up to 14% in males and 11% in females. The result was replicated across multiple independent laboratories simultaneously, instantly making it one of the most significant findings in longevity research.

How Does Rapamycin Work? Understanding mTOR Inhibition
To understand rapamycin's effects, you first need to understand its molecular target: mTOR, or the mechanistic target of rapamycin.
mTOR is a master regulatory kinase — essentially a central control switch for cell growth, metabolism, and aging. When nutrients are plentiful and conditions are favorable, mTOR drives the cell to grow, produce proteins, and divide. This is necessary during development, but as we age, chronically elevated mTOR activity drives cellular dysfunction and accelerates aging.
Rapamycin works by binding to an intracellular protein called FKBP12, forming a complex that inhibits mTOR complex 1 (mTORC1). This seemingly simple molecular interaction has cascading biological effects — many of them profoundly beneficial.
Autophagy: Cellular Housekeeping Restored
One of the most therapeutically important consequences of mTOR inhibition is the activation of autophagy — the cell's intrinsic mechanism for breaking down and recycling damaged proteins and worn-out organelles.
As we age and mTOR remains chronically active, autophagy declines, and cellular debris accumulates. This is particularly damaging in neurons and heart cells, which cannot regenerate by division. Rapamycin releases the mTOR-imposed suppression of autophagy, restoring the cell's ability to clean itself — a fundamental anti-aging effect at the cellular level.
Senescence and SASP Suppression
Cellular senescence refers to the state in which aging cells permanently exit the cell cycle but remain metabolically active, secreting a damaging cocktail of inflammatory signals known as the senescence-associated secretory phenotype (SASP). mTORC1 activity is required to maintain SASP, meaning rapamycin — by inhibiting mTORC1 — directly reduces this chronic inflammatory output from senescent cells. The result is less "bystander damage" to surrounding healthy tissue.
Rapamycin and Lifespan Extension: The Evidence Across Species
The lifespan data for rapamycin is among the most consistently replicated findings in modern biology.
Invertebrate Models
In Caenorhabditis elegans (roundworms), rapamycin extends lifespan by 20–30% depending on dosing. Comparable results have been demonstrated in Drosophila melanogaster (fruit flies) and the yeast Saccharomyces cerevisiae. The fact that TOR pathway inhibition extends life across organisms separated by hundreds of millions of years of evolution strongly suggests this is a deeply conserved mechanism of longevity — not a quirk of any single species.
Rodent Models
In mice, subsequent ITP studies building on the 2009 findings achieved lifespan extensions of up to 23% in males and 26% in females with earlier treatment initiation. Critically, rapamycin-treated mice did not merely live longer — they experienced delayed onset of multiple age-related diseases including cardiac hypertrophy, liver lesions, and physical decline. This compression of morbidity — more healthy years, not just more years — is precisely what longevity medicine aims for.
Companion Animal Studies
The Dog Aging Project TRIAD Trial has been evaluating rapamycin in middle-aged companion dogs — animals that share human environments and develop strikingly similar age-related diseases. Early echocardiographic results showed measurable improvements in cardiac structure and function in treated dogs, representing a meaningful translational step between rodent models and human biology.
Rapamycin and Immune Rejuvenation: The Clinical Evidence
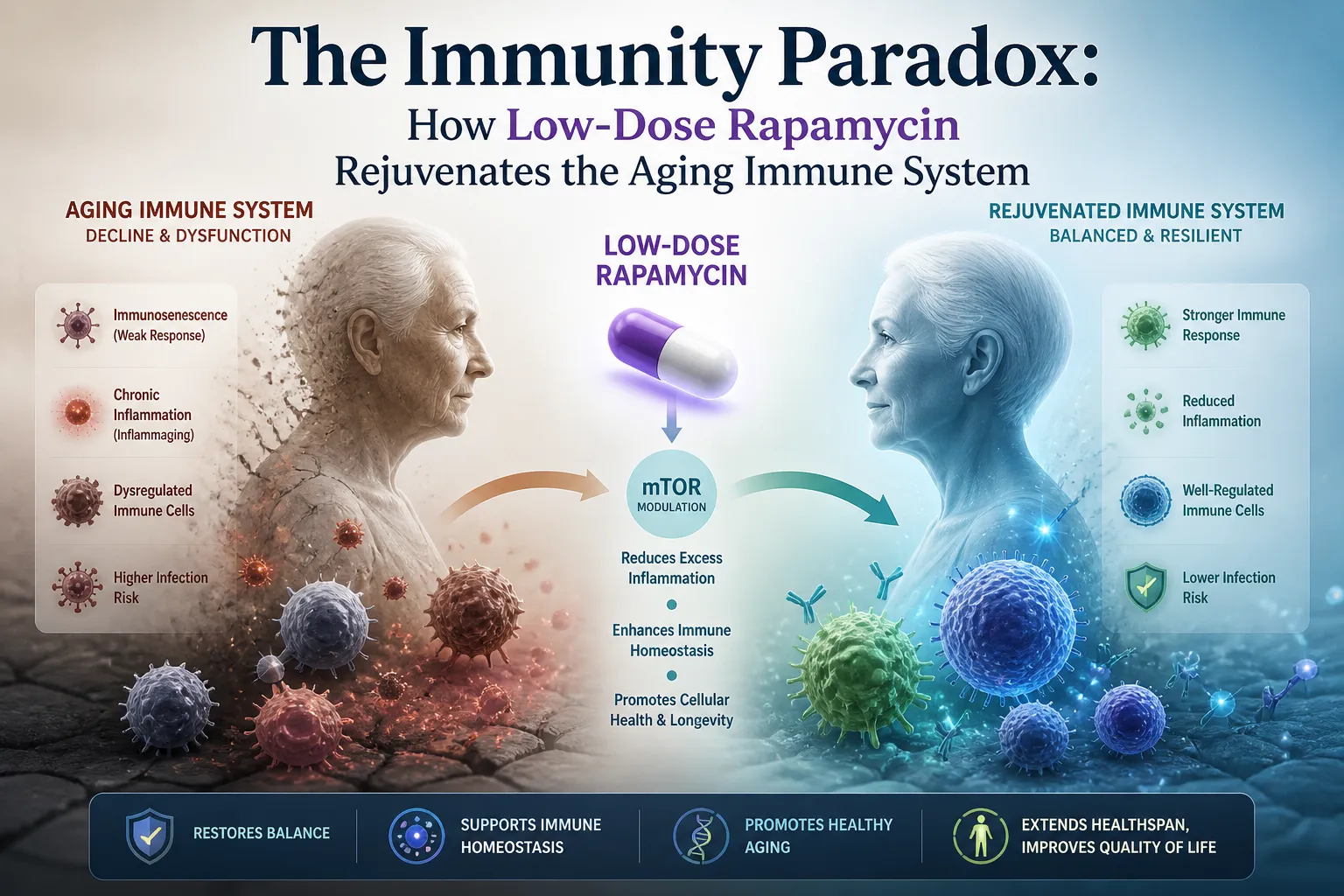
Perhaps the most surprising chapter in rapamycin's story is what it does to the aging immune system.
Aging is accompanied by immunosenescence — a progressive decline in immune function characterized by a shrunken naive T cell repertoire, accumulation of exhausted memory cells, and chronic low-grade inflammation. Vaccine responses weaken. Infection rates rise. The immune system that once protected us efficiently begins to falter.
In a pivotal clinical trial, Dr. Joan Mannick and colleagues administered a rapamycin analog (everolimus/RAD001) to healthy elderly volunteers for six weeks prior to seasonal flu vaccination. The results were striking:
- Vaccine response improved by 20% compared to placebo
- The proportion of exhausted PD-1-positive T cells decreased significantly
- The ratio of naive-to-memory T cells — a hallmark of a younger immune system — increased
This was not immunosuppression. This was measurable, functional immune rejuvenation.
A follow-up 2018 study confirmed the findings, showing reduced infection rates and improved self-reported health in treated participants. The emerging model — sometimes called "immune rheostatting" — suggests that low-dose intermittent rapamycin recalibrates aged immune systems toward a more youthful functional state, rather than broadly suppressing immunity.
Neuroprotection: Rapamycin and Brain Health
The central nervous system presents one of the most compelling cases for rapamycin's benefits. Neurons are post-mitotic — they cannot replace themselves through division — making them uniquely dependent on autophagy to clear accumulated protein damage over decades.
Alzheimer's Disease
In transgenic mouse models of Alzheimer's disease, rapamycin treatment has consistently:
- Reduced amyloid plaque burden
- Decreased tau pathology
- Improved spatial memory performance in behavioral testing
- Rescued cognitive deficits even when started late in life
This last point is particularly significant. It suggests a therapeutic window that extends well into old age — not a drug that must be started in youth to have any benefit.
Parkinson's Disease
In Parkinson's disease models, rapamycin-induced autophagy promotes clearance of aggregated alpha-synuclein — the protein whose accumulation drives dopaminergic neuron death. Since conventional proteasomal degradation cannot process large protein aggregates, autophagy is the only viable clearance route. Rapamycin opens and sustains that route.
Normal Cognitive Aging
Beyond disease models, rapamycin has been shown to reverse age-related transcriptional changes in the hippocampus of normal aging mice — including restoration of genes linked to synaptic plasticity — correlating with preserved performance on hippocampal-dependent memory tasks.
Cardiovascular Benefits: Protecting the Aging Heart
Cardiovascular disease remains the leading cause of death in aging populations, and the heart is among the organs most profoundly affected by chronic mTOR overactivation.
Reversing Cardiac Aging
In aged mice, rapamycin treatment:
- Reduced age-associated cardiac hypertrophy (thickening of the heart muscle)
- Improved diastolic function
- Shifted gene expression profiles in aged hearts toward more youthful patterns
One laboratory demonstrated that a short course of rapamycin in old mice produced cardiac improvements that persisted after the drug was discontinued — suggesting durable structural or epigenetic changes, not merely transient suppression.
Vascular Health
Rapamycin analogs are already the active ingredient in drug-eluting coronary stents, where they prevent restenosis (artery re-narrowing) by inhibiting vascular smooth muscle cell proliferation. This constitutes decades of real-world clinical safety data for rapamycin's vascular effects. Preclinical atherosclerosis models show that systemic rapamycin also reduces plaque burden and improves arterial compliance through anti-inflammatory and anti-senescent mechanisms.
Metabolic Health and Body Composition
mTOR signaling is deeply embedded in metabolic regulation, and rapamycin's modulation of this pathway produces several metabolically favorable outcomes.
Rapamycin has been shown to reduce visceral fat accumulation in rodent aging models by suppressing mTORC1-driven lipogenesis and promoting fat oxidation. Mitochondrial biogenesis — essential for cellular energy metabolism — is also supported through upregulation of PGC-1α activity secondary to mTORC1 inhibition.
An important caveat: chronic high-dose rapamycin can impair insulin sensitivity through disruption of mTORC2, which plays a role in insulin signaling. This is a key reason why intermittent dosing protocols — such as once-weekly administration — are increasingly favored in longevity research. Intermittent dosing achieves the benefits of mTORC1 inhibition while largely sparing mTORC2 activity and preserving metabolic homeostasis.
Cancer Prevention: A Compelling Case
mTOR hyperactivation is present in the vast majority of human cancers — essentially a growth engine that tumors exploit. Rapamycin analogs (temsirolimus, everolimus) already hold FDA approval for treating renal cell carcinoma, certain breast cancers, and pancreatic neuroendocrine tumors.
From a preventive standpoint, ITP mouse data show that rapamycin-treated animals not only live longer but display a reduced incidence of late-life malignancies. The cancer-preventive mechanisms are multiple: suppression of aberrant proliferation, enhanced immune surveillance, reduction of the SASP-driven pro-tumor microenvironment, and autophagy-mediated clearance of genomically damaged pre-malignant cells.
Importantly, the immunosuppressive concern does not appear to apply at longevity-appropriate low doses — where immune function is in fact enhanced, not suppressed.
Addressing Safety: What You Need to Know
Any honest discussion of rapamycin must address its side effects.
At the high continuous doses used in transplant medicine, rapamycin is associated with impaired wound healing, mucositis, elevated triglycerides, and potential metabolic effects. These are real and clinically significant in that context.
However, the longevity research community is exploring a fundamentally different approach:
- Lower doses (typically 1–6 mg versus transplant doses of 5–15 mg daily)
- Intermittent administration (most commonly once weekly)
- Defined treatment courses rather than indefinite daily use
At these intermittent lower doses, mTORC1 is inhibited during the dosing window while mTOR activity recovers between doses — preserving mTORC2 function, maintaining metabolic health, and avoiding the cumulative toxicity of continuous high-dose regimens.
The Mannick clinical trials used six-week courses rather than indefinite daily treatment, and demonstrated immune benefits without significant adverse effects. This does not mean rapamycin is without risk — it remains a prescription drug requiring medical supervision — but it does mean that the risk-benefit calculation at longevity doses is substantially more favorable than transplant-dose experience would suggest.
Emerging Clinical Trials: What to Watch
The clinical pipeline for rapamycin as a geroprotective agent is rapidly expanding:
The PEARL Trial (Participatory Evaluation of Aging with Rapamycin for Longevity) is assessing the effects of rapamycin on biological aging biomarkers — including epigenetic clocks, inflammatory panels, and immune phenotyping — in healthy older adults. This may provide the first rigorous human data on whether rapamycin moves validated measures of biological age.
The Dog Aging Project TRIAD Trial is generating organ-level outcome and longevity data in companion dogs — a large mammalian model with significantly more translational relevance to human aging than rodents.
Researchers are also actively investigating potential synergies between rapamycin and other geroprotective interventions including caloric restriction, metformin, and senolytic drugs that selectively eliminate senescent cells.
Conclusion: A New Paradigm in Aging Medicine
Rapamycin stands apart in the landscape of longevity research for one simple reason: the breadth, consistency, and mechanistic depth of its supporting evidence is unmatched by any comparable compound.
From lifespan extension across multiple species, to measurable immune rejuvenation in human clinical trials, to neuroprotection, cardiovascular preservation, and cancer prevention — the benefits of appropriately dosed rapamycin are numerous, interconnected, and grounded in well-understood biology.
We are at an early but genuinely promising moment in the translation of rapamycin from transplant immunosuppressant to candidate longevity medicine. The ongoing clinical trials will be decisive in determining how fully this promise translates to human benefit.
But based on the evidence available today, rapamycin deserves to be part of every serious conversation about the future of healthy aging.

Frequently Asked Questions (FAQs)
Q: Is rapamycin safe to take for anti-aging purposes? A: At the low intermittent doses being explored in longevity research (typically once weekly at lower doses), rapamycin appears to have a more favorable safety profile than the high-dose continuous transplant regimen. However, it remains a prescription drug and should only be used under medical supervision.
Q: How is rapamycin different from other anti-aging supplements? A: Unlike most supplements, rapamycin has a well-characterized molecular mechanism (mTOR inhibition), decades of clinical use, and robust preclinical lifespan data across multiple species. It is a pharmaceutical drug, not a supplement.
Q: Does rapamycin suppress the immune system? A: At high continuous doses used in transplant medicine, yes. But clinical trials have shown that at low intermittent doses, rapamycin can actually improve immune function in elderly individuals, increasing vaccine response and reducing T cell exhaustion.
Q: What is mTOR and why does it matter for aging? A: mTOR (mechanistic target of rapamycin) is a master regulatory protein that controls cell growth and metabolism. Chronically elevated mTOR activity is associated with accelerated aging. Reducing mTOR activity — as rapamycin does — is one of the most consistently effective interventions for extending healthy lifespan in animal models.
Q: Are there human clinical trials for rapamycin and aging? A: Yes. The PEARL trial and other studies are currently investigating rapamycin's effects on biological aging markers in healthy older adults. The Dog Aging Project is also providing important translational data.

















Sign in to leave a comment.